-
iestinstrument
Analysis of Gas Production Behavior Of LFP Cells During Overcharge And Overdischarge Stages
1. Preface
Lithium iron phosphate (LFP cells), which typically use LiFePO₄ with an olivine structure coated onto aluminum foil as the cathode and graphite coated onto copper foil as the anode, have become the preferred choice for new energy vehicles and energy storage power stations due to their superior safety profile. During charging, Li⁺ ions migrate from the LiFePO₄ particle surface into the electrolyte, pass through the separator, and embed into the graphite lattice on the anode side, forming an intercalation compound (LiCₓ). Concurrently, electrons flow from the aluminum foil current collector through the external circuit to the copper foil, maintaining charge balance.
As Li+ ions are extracted, the cathode material transforms from LiFePO₄ to Li1-xFePO4. Discharge involves the reverse process: Li+ ions deintercalate from the graphite anode, move through the electrolyte and separator, and re-embed into the LiFePO₄ lattice. Electrons flow externally from the copper foil to the aluminum foil, ensuring charge equilibrium. Figure 1 illustrates the working principle of an LFP cell.
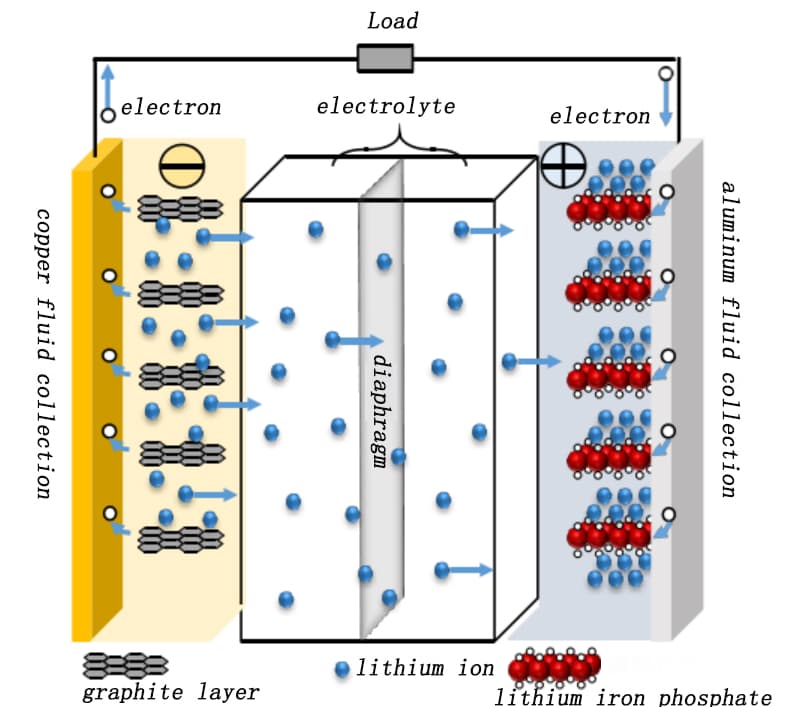
Figure 1. Schematic diagram of the working principle of the LFP full electric cell [1]
In practical applications, both overcharge and overdischarge can cause severe damage to the cell and significantly reduce its lifespan. Overcharging may lead to lithium plating and gas generation, while overdischarging can result in copper dendrite formation and gas evolution—both of which accelerate performance degradation and may even cause thermal runaway. This study uses an in-situ gas & volume monitor(GVM) combined with gas chromatography to perform real-time analysis of battery gases produced by LFP cells during controlled overcharge and overdischarge tests. The goal is to quantify gas evolution, identify gas species, and correlate gas production with electrochemical events to support failure analysis and safety design.
2. Experimental Equipment and Test Methods
2.1 Instruments
GVM2200 — In-situ Battery Gassing & Volume Analyzer (IEST)
- Temperature range: 20 °C – 85 °C.
- Dual-channel synchronous testing (two cells).
- Real-time logging of cell volume, temperature, current, voltage and capacity.
(Figure 2 — GVM2200 appearance)
Gas Chromatography (GC-2014C) for gas analysis battery:
- Samples taken (1 mL) from the cell headspace inside an inert glovebox.
- Detectors: TCD (thermal conductivity) and FID (flame ionization) to cover H₂, CO, CO₂, CH₄, C₂H₆, C₂H₂, and other hydrocarbons.
Figure 2. Appearance diagram of the GVM2200 equipment
2.2 Overcharge & Overdischarge Protocols
-
Cells preconditioned to 2.5 V, hold 2 h.
-
Cell A (overcharge): 0.5 C (1.5 A) CCCV to 5.0 V, cutoff current 0.2 mA, hold.
-
Cell B (overdischarge): 0.5 C (1.5 A) DC discharge to 0 V, hold.
-
Initial mass (m₀) recorded; GVM2200 records continuous volume changes while GC samples headspace gas for analysis battery gases.
2.3 Test Method
Each cell was weighed initially (m₀) and placed into the GVM2200. Using the MISG software, parameters including cell ID and sampling frequency were set. The system automatically recorded volume change, temperature, current, voltage, and capacity. For gas analysis, 1 mL of gas was extracted in a glove box and analyzed using a GC-2014C gas chromatograph with TCD and FID detectors. Detectable gases are shown in Figure 3.

Figure 3. Gas composition that can be tested by FID and TCD detectors
 3. Analysis of the Results of Volume Change and Gassing Behavior
3. Analysis of the Results of Volume Change and Gassing Behavior
3.1 Overcharge, Overcharge and Discharge, and Volume Change Curve Analysis
As shown in Figure 4, the volume and voltage change curve of the lithium ion from the positive electrode during the normal charging stage of the charging cell, the voltage increases, as the voltage increases, and the cell volume increases, and the process of graphite can reach 10%[2]。Graphite negative electrode is a typical phased interlayer lithium embedding process. After lithium ion is embedded, the layer keeps the plane. The graphite layer and the embedded layer are arranged in parallel, and every third layer, 2 and one layer are regularly embedded to form Li-C interlayer compounds (LiCx) with different phases such as 3,2 and 1.The initial stage is stage 4, and the state of each three layer of lithium ion is called stage 3, which corresponds to Li0.3C6Compounds, with a relative lithium concentration of 33.33%.Each two layers of lithium embedding is stage 2, corresponding to Li0.5C6, The relative concentration was 50%.After the graphite is completely embedded with lithium, the LiC is formed6Compound, one lithium ion embedded in the middle of every six hexagonal carbon atoms, is a 100% relative lithium-embedded concentration[2].
As shown in Figure 5 shows the change of the negative state in the normal charging stage of LFP cell. The above lithium embedded stage is in a completely ideal state. The actual lithium embedded state inside graphite is more complex, which is often a mixture of multiple stages.The volume change of the corresponding cell charging stage is mainly related to the structural phase change caused by the negative electrode lithium embedding[5], In the initial stage of charging, with the increase of lithium embedded, the volume of graphite lattice expands, forming the expansion curve with the larger slope of the first stage, the lattice size of graphite changes least between x=0.2 and 0.6, and the expansion appears a platform curve; LiC6The layer spacing of the phase is significantly greater than that of Li0.5C6each other.equal LiC6The maximum slope for the increase of the corresponding thickness change occurs in the presence of the phase.
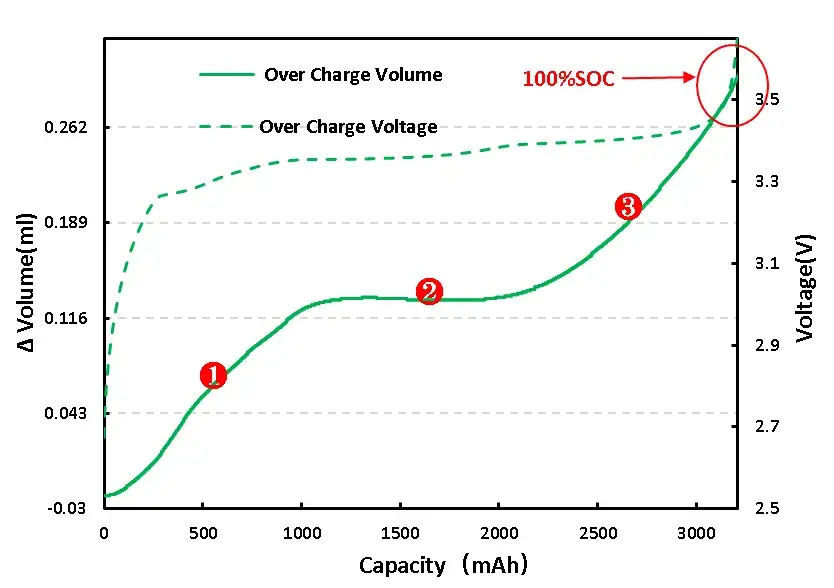
Figure 4. Volume change in the normal charging stage of the overcharging cell
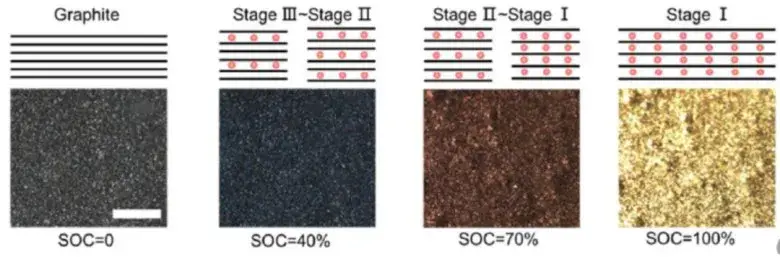
Figure 5. (1)Changes in graphite negative electrode state during normal charging of lithium-ion batteries[2]
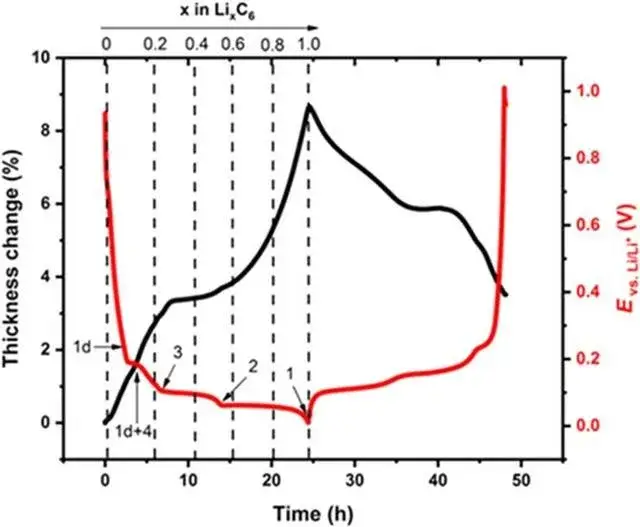
Figure 5. (2)Graphite volume expansion curve[5]
3.2 Overcharge and Overdischarge Volume Signatures
-
Overcharge (to 5.0 V): Volume vs. voltage curves show an inflection point near the overcharge onset (~110% SOC), and sustained volume growth follows even while the voltage remains at the overvoltage plateau. Visible pouch bulging occurs, indicating gas generation and irreversible swelling (Figure 6a).
-
Overdischarge (to 0 V): Early volume is stable, but below ~0.4 V an inflection appears; prolonged low-voltage conditions drive continued volume increase and mild bulging (Figure 6b).
These volume signatures enable rapid detection of gas onset and provide timing for GC sampling for gas analysis battery.
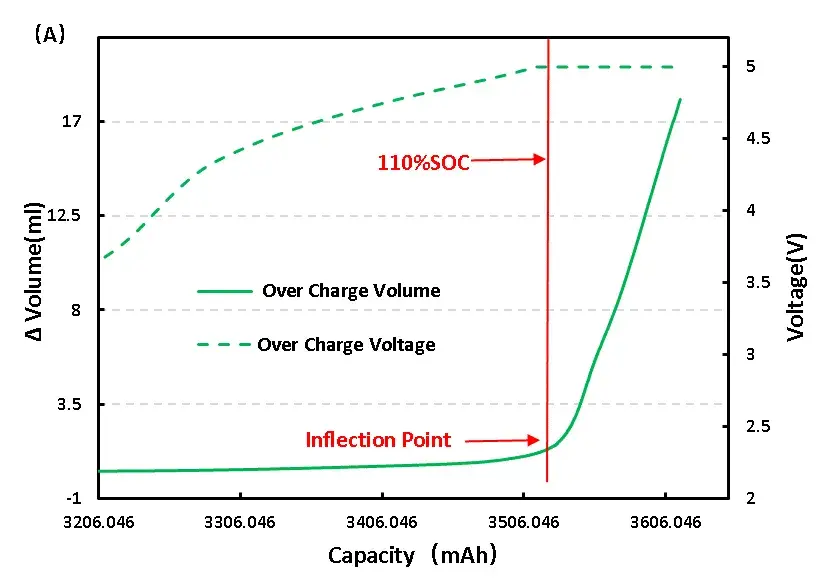
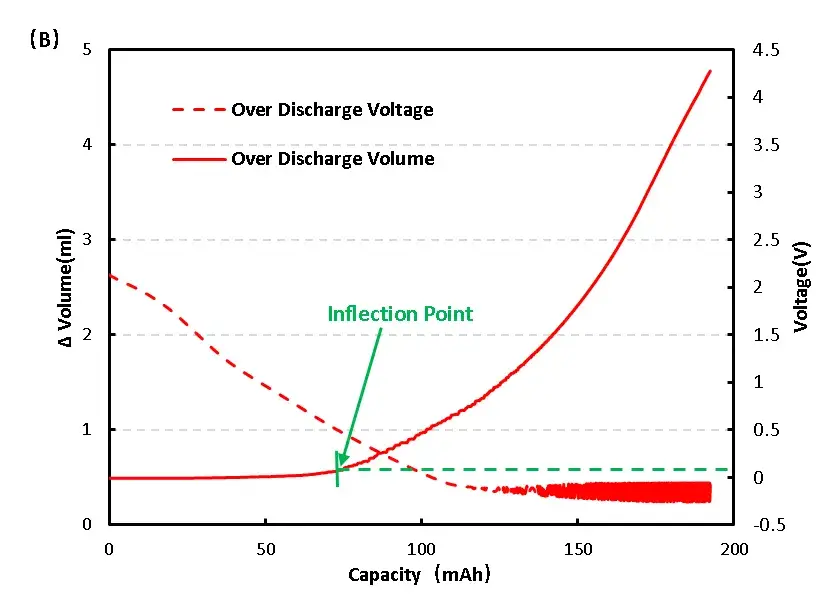
Figure 6. Change of overcharge and overdischarge volume of LFP cell
4. Gas Composition — Analysis of Battery Gases
4.1 GC Results Overview
Headspace samples taken after overcharge/overdischarge were analyzed by GC (TCD/FID). Major findings:
-
Hydrogen (H₂): High proportion in both overcharge and overdischarge cases — likely from trace water decomposition or side reactions that release H₂. Moisture impurities in electrodes or electrolyte can catalyze H₂ evolution at elevated potentials/temperatures.
-
Carbon Oxides (CO, CO₂): Detected in substantial quantities during overcharge, indicating oxidative decomposition of electrolyte solvents (carbonate solvents) and organic components. CO₂ is a common decomposition product of carbonate electrolyte oxidation.
-
Light Hydrocarbons (CH₄, C₂H₆, C₂H₂, etc.): Present in smaller but measurable amounts, reflecting complex electrolyte breakdown pathways and possible solvent/SEI degradation.
(Figures 7–8 and Table 1 summarize GC spectra and quantitative comparisons of gas species for overcharge vs. overdischarge.)
4.2 Interpretation of Gas Origins
-
H₂: May originate from water desorption/decomposition on electrodes and catalytic hydrogen evolution at high potentials. Literature suggests water desorption under vacuum at ~350 K with activation energy ~1.3 eV; in cells, localized heating and catalysis accelerate H₂ production.
-
CO / CO₂: Oxidative decomposition of carbonate solvents (EC, DMC, EMC, DEC) at high potentials generates COx species. Overcharge to very high voltages amplifies solvent oxidation and CO₂ formation.
-
Hydrocarbons / C₂ species: Radical pathways and electrolyte fragmentation produce small hydrocarbons; presence of C₂H₂ may signal severe electrolyte breakdown.
Overall, gas analysis battery results confirm that overcharge produces a richer mixture of oxidative decomposition gases (CO, CO₂, hydrocarbons) in addition to H₂, while overdischarge yields significant H₂ and lower levels of oxidation products.
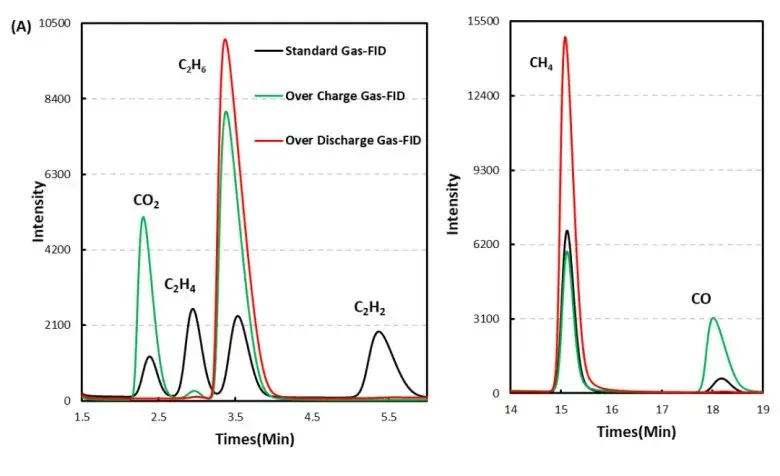
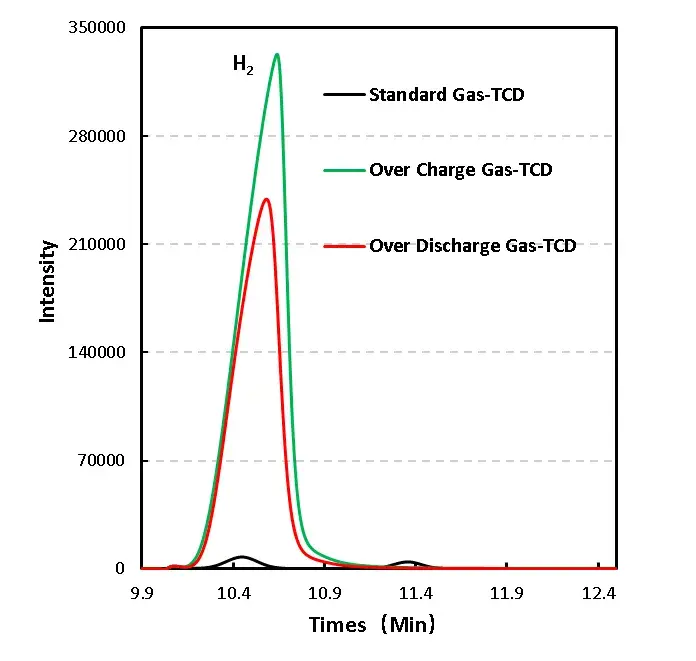
Figure 7. GC test results of overcharged and overdischarged gas production components of LFP cells
Table 1. Comparison Table of Overcharge and Overproduction Components of LFP Cells
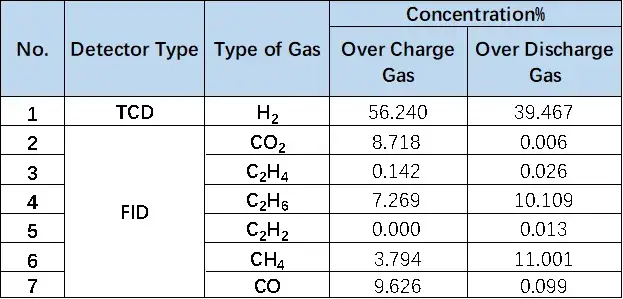
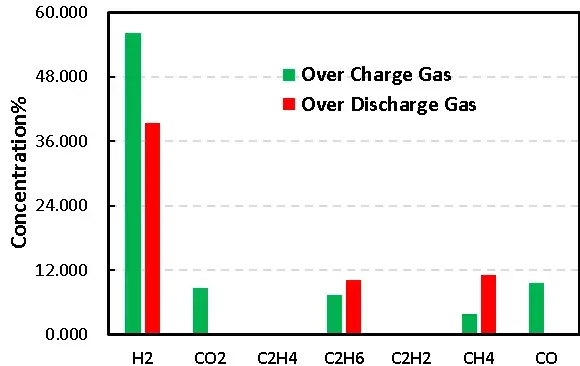
Figure 8. Comparison of overcharge and overdischarge gas production of LFP cells
5. Discussion — Implications for LFP Cell Safety and Diagnostics
-
Early detection: Real-time volume monitoring (GVM Series) provides fast indicators of gas generation, enabling timely intervention or shutoff during testing and manufacturing. Coupling volume signals with targeted GC sampling yields robust analysis battery gases results.
-
Source control: Minimizing residual moisture and electrolyte impurities is critical to reduce H₂ evolution. Strict material drying and dry-room controls lower gas hazards.
-
Electrolyte design: Electrolyte formulations and additives that improve oxidative stability can reduce CO/CO₂ formation under overcharge. LFP cells, while safer than high-Ni chemistries, still experience solvent oxidation at extreme states.
-
Failure forensics: Quantitative gas species and timing relative to SOC/voltage allow root-cause analysis — distinguishing thermal runaway precursors from purely electrochemical decomposition.
6. Summary
Combining in-situ gassing volume monitoring (GVM2200) with gas chromatography provides a powerful gas analysis battery workflow to study LFP cell gassing under abuse conditions. Key outcomes:
-
Overcharge produces a mix of gases dominated by H₂, CO, and CO₂, plus light hydrocarbons — consistent with electrolyte oxidation and water-related reactions.
-
Overdischarge also shows significant H₂ production, reflecting moisture-related and reductive side reactions.
-
Real-time volume trends allow precise timing for GC sampling and support rapid R&D diagnostics, safety validation, and quality control for LFP-based systems.
This combined approach helps engineers and researchers better understand gas generation mechanisms, improve cell formulations, and design safer batteries and packs.
6. References
[1] Zheng Zhikun Cheng.Research on lithium iron phosphate energy storage overcharged thermal runaway and gas detection safety early warning [D].Zhengzhou University.
[2] Reynier Y, Yazami R, Fultz B, et al. Evolution of lithiation thermodynamics with the graphitization of carbons[J].Journal of Power Sources, 2007, 165(2):552-558.
[3] Yang L, Chen H S, Song W L, et al. Effect of Defects on Diffusion Behaviors of Lithium-Ion Battery Electrodes: In Situ Optical Observation and Simulation[J].ACS Applied Materials & Interfaces, 2018, 10(50).
[4] Kajiura H, Nandyala A, Bezryadin A. Quasi-ballistic electron transport in as-produced and annealed multiwall carbon nanotubes[J].Carbon, 2005, 43(6):1317-1319.
[5] H.Michael, F.Iacoviello, T.M.M.Heenan, A.Llewellyn,J.S.Weaving, R.Jervis, D.J.L.Brett, and P.R.Shearing. A Dilatometric Study of Graphite Electrodes during Cycling with X-ray Computed Tomography[J]Journal of the Electrochemical Society, 2021,168: 010507.
Contact Us
If you are interested in our products and want to know more details, please leave a message here, we will reply you as soon as we can.



