-
iestinstrument
Research Status of All Solid-state Batteries
1. Preface
Lithium-ion batteries stand out from many other energy storage technologies due to their advantages of high energy density, long cycle life, low self-discharge rate, no memory effect, etc., and have become the most important part of the current electrochemical energy storage field. In recent years, the application scenarios of lithium-ion batteries have been rapidly broadening, from portable electronic products to electric vehicles, home energy storage, industrial energy storage, etc., lithium-ion batteries have penetrated into all aspects of society, which also puts forward higher requirements for the safety and energy density of lithium-ion batteries.
Traditional lithium-ion batteries rely on organic liquid electrolyte to conduct lithium ions, and the flammable and volatile nature of the electrolyte leads to thermal runaway, fire, explosion, and other safety risks when the battery is overheated, short-circuited, overcharged, or mechanically damaged. New energy vehicle safety accidents occur from time to time, to ensure the safe operation of new energy vehicles is the primary task of the current industry development. In recent years, through the battery system structure innovation, to a certain extent, to improve the power battery operation safety. Looking to the future, from the battery structure innovation to material system innovation is the inevitable trend of power battery.
In addition, energy density is a key indicator of power lithium batteries. The “Made in China 2025” and “New Energy Vehicle Industry Development Plan (2021~2035)” and other plans propose that by 2025 and 2030, the single energy density of the electric core will gradually realize the goal of 400~500Wh/kg. However, liquid lithium-ion batteries are limited by the chemical system, and the upper limit of energy density is expected to be 300Wh/kg, which is difficult to meet the future strategic demand.
Therefore, the development of high safety and high energy density energy storage technology to make up for the shortcomings of liquid lithium-ion batteries is an important development direction for future lithium-ion batteries.
The use of solid-state electrolytes to replace liquid electrolytes and the development of all solid-state batteries can solve the energy density bottleneck and safety hazards faced by current liquid ion batteries, and become the most promising next-generation lithium-ion battery successor technology. As shown in Figure 1, the structure and working principle of all solid-state batteries are similar to that of liquid lithium-ion batteries, but the solid-state electrolyte has higher thermal and chemical stability, and will not leak, burn or explode, thus reducing the risk of thermal runaway and improving the intrinsic safety of the battery. At the same time, the solid-state electrolyte has a high Young’s modulus, which can effectively inhibit the growth of lithium dendrites, and is expected to realize the application of lithium metal anode, so that the energy density of the battery is greatly increased. In addition, the packaging of all solid-state batteries is easier. The use of bipolar stacking technology in all solid-state batteries can reduce the use of inactive materials such as lugs and leads, and the battery module does not require a cooling system, which is expected to further improve the volume and mass energy density of the system.
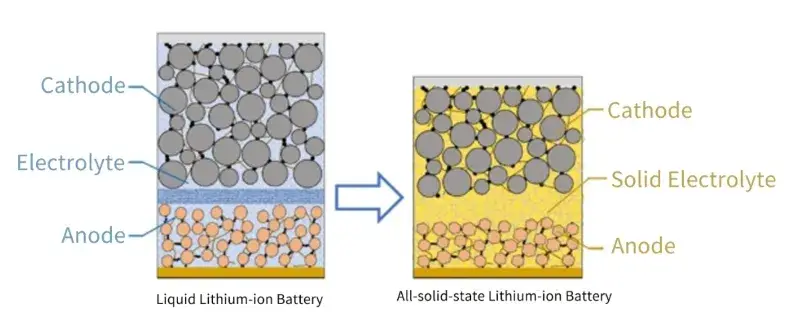
Figure 1. Comparison of the structure of liquid lithium-ion batteries and all solid-state lithium-ion batteries
2. Current Status of Solid Electrolyte Research
Solid electrolyte is the core material of all solid-state batteries and is also known as a fast ion conductor. It refers to a type of material that is solid within the operating temperature range, exhibits good insulation against electrons, and has high lithium-ion conductivity. Due to its certain mechanical strength and ability to quickly conduct lithium ions, solid electrolytes can effectively replace the electrolyte and separator in commercial lithium batteries and become the medium that provides lithium-ion transmission paths inside all solid-state batteries.
Currently, researchers have developed a variety of solid electrolytes. According to their composition, solid electrolytes can be divided into three categories: polymers, inorganics and organic/inorganic composite electrolytes. Among them, inorganic solid electrolytes can be divided into subdivided types such as oxides and sulfides.
2.1 Polymer Solid Electrolyte
Polymer-based solid electrolytes are generally solid solutions formed by uniform mixing of polymer matrix and lithium salt. Compared with inorganic solid electrolytes, polymer solid electrolytes have the advantages of good flexibility, high viscosity, low price, and easy processing. Polar groups in the polymer electrolyte, such as C=O, C=N, -O-, -S-, etc., can coordinate with lithium ions, thereby promoting the dissolution of lithium salts and producing freely moving ions. At present, most studies believe that ion transport in polymer electrolytes mainly occurs in the amorphous region above Tg, so the mobility of its chain segments is also the key to efficient ion transport. As shown in Figure 2, lithium ions coordinate with polar groups on the polymer chain at specific positions, and through local segmental movement of the polymer chain, free volume is generated, allowing lithium ions to conduct within and between chains.
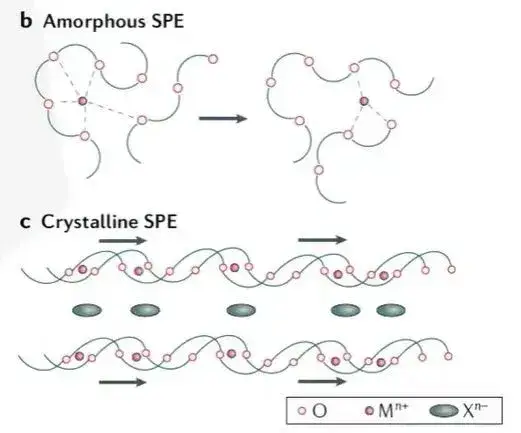
Figure 2. Schematic diagram of the ion transport mechanism of polymer solid electrolyte
Polymer solid electrolytes usually have low room temperature ionic conductivity. For example, the ionic conductivity of PEO is in the range of 10-7~10-5 S/cm, which is difficult to meet the actual use needs of all solid-state batteries. To improve the ionic conductivity of polymer electrolytes, common methods include adding plasticizers to increase the free volume between segments or adding organic solvents to further promote the dissolution of lithium salts.
2.2 Inorganic Solid Electrolyte
Ion migration in crystalline materials generally follows the Arrhenius formula, which depends on the concentration and distribution of defects in the crystal structure. As shown in Figure 3, the ion diffusion mechanism based on Schottky and Frenkel point defects includes simple vacancy diffusion and relatively complex double vacancy mechanism, gap mechanism, gap-displacement exchange mechanism, etc.
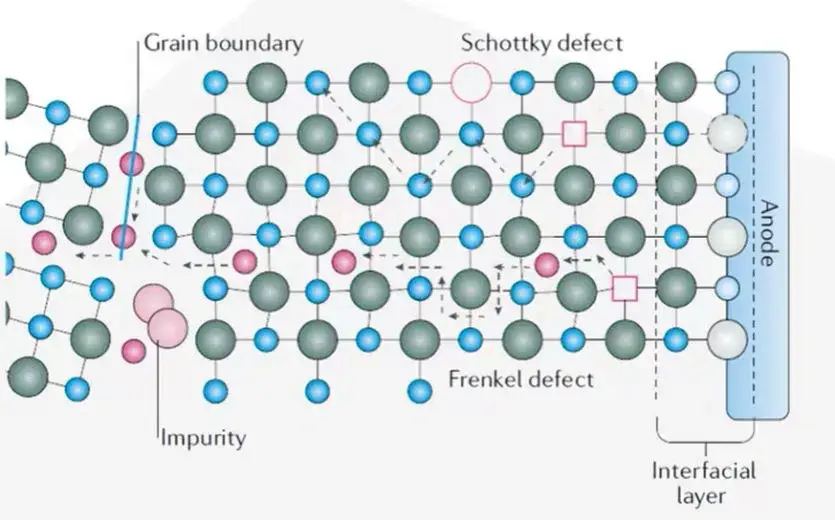
Figure 3. Schematic diagram of the ion transport mechanism of crystalline solid electrolyte
2.2.1 Oxide Solid Electrolyte
Oxide solid electrolytes can be mainly divided into perovskite type Li5La3Ti2O12 (LLTO), garnet type Li7La3Zr2O12 (LLZO), LISICON type Li14Zn (GeO4)4 and NASICON type Li1.5Al0.5Ge1.5(PO4)3 (LAGP), Li1.3Al0.3Ti1.7(PO4)3 (LATP) solid electrolyte. The room temperature ionic conductivity of oxide solid electrolytes is in the range of 10-4~10-3 S/cm. The conductivity of oxide solid electrolytes can be further improved through element doping, optimized preparation processes and other means. For example, doping LLZO with Ta, Al or Ca can increase the lithium holes in the crystal and increase its conductivity to a level of 10-3 S/cm. In addition, ultra-fast sintering technology can avoid the loss of raw materials during the sintering process and synthesize high-quality ceramics.
Different oxide solid electrolytes exhibit differentiated chemical/electrochemical stability due to different crystal structures and elemental compositions. When LATP and LAGP are matched with lithium metal anodes, high-valent ions Ti4+ and Ge4+ will be reduced by metallic lithium to form an interface intermediate phase with mixed conductivity of ions and electrons, which directly affects the stability of the interface. Although LLZO electrolyte is theoretically chemically stable in the air, studies have found that LLZO easily reacts with moisture and CO2 in the air to form a harmful Li2CO3 passivation layer on the surface, thereby affecting the ionic conductivity of the electrolyte and the interface with the metallic lithium anode wettability.
The Young’s modulus of the oxide solid electrolyte is relatively high, which can theoretically effectively inhibit the growth of lithium dendrites. However, a high Young’s modulus is also not conducive to the physical contact between the electrolyte and the electrode interface, and cannot effectively accommodate the volume change of the electrode material during the electrochemical process, as a result, the local stress generated inside the all solid-state batteries cannot be effectively released, causing contact loss and mechanical failure of the battery. In addition, the brittleness of oxide solid-state electrolytes is high, and high-temperature annealing processes are usually required when preparing ceramic sheets and composite cathodes, which makes the preparation of corresponding types of all solid-state batteries a considerable technical challenge. On the one hand, it is necessary to ensure that the electrolyte does not undergo brittle fracture during the preparation process of the all solid-state batteries; on the other hand, it is necessary to ensure that the electrolyte and the positive electrode do not undergo violent diffusion of elements during the co-sintering process to avoid phase changes in the material structure. Therefore, introducing a polymer buffer layer at the interface between the electrolyte and the electrode to construct a multilayer electrolyte structure or preparing an organic-inorganic composite solid electrolyte can effectively solve the physical incompatibility between the oxide electrolyte and the electrode, and is conducive to its large-scale production and preparation
2.2.2 Sulfide Solid Electrolyte
Compared with oxide solid electrolytes, sulfide ions have a larger radius and smaller electronegativity than cations, so sulfide solid electrolytes have a lower binding force to lithium ions. This makes the ion transport channel formed in the sulfide wider than the oxide electrolyte and transports lithium ions more easily, thus showing higher ionic conductivity. In addition, sulfide solid electrolytes also have the advantages of excellent thermal stability and good mechanical properties. Based on composition and structure, sulfide solid electrolytes are mainly divided into glassy, Thio-LISICON, glass-ceramic, lithium sulfide silver germanium ore type, and LGPS superionic conductors. Glassy sulfide solid electrolytes include Li2S-GeS2、Li2S-SiS2、Li2S-P2S5-LiI, and their room temperature lithium ion conductivity is ~10-4 S/cm. Thio-LISICON type Li3.25Ge0.25P0.7S4 room temperature lithium-ion conductivity is 2.2×10-3 S/cm. Glass-ceramic Li7P3S11 room temperature lithium-ion conductivity is 2.2×10-3 S/cm. The lithium sulfide-silvergermanium sulfide electrolyte Li6PS5X (X=Cl, Br, I) has high lithium-ion conductivity, and the room temperature conductivity of Li6PS5Cl synthesized by the solid-phase method is as high as 4.96×10-3 S/cm. The superionic conductor Li10GeP2S12 (LGPS) is considered a milestone, with a room temperature lithium-ion conductivity of 1.2×10-2 S/cm, which is comparable to liquid electrolytes. After halogen doping, the room temperature lithium-ion conductivity of Li9.54Si1.74P1.44S11.7Cl0.3 superionic conductor even exceeds that of liquid electrolyte, reaching 2.5×10-2 S/cm.
In addition to high ionic conductivity, the unique mechanical properties of sulfide electrolytes are also one of the reasons why they are so popular. The modulus of sulfide electrolytes is between that of oxides and polymers. From the perspective of material mechanics, sulfide electrolytes can not only inhibit the growth of lithium dendrites, at the same time, its texture is soft and can be densified through simple cold pressing, avoiding the high-temperature sintering process similar to oxide ceramics. However, sulfide electrolytes have low fracture toughness values and are prone to brittle fracture during battery preparation and cycling, causing mechanical failure of the battery.
Most sulfide solid electrolytes are unstable in the air. A small amount of moisture in the air can cause hydrolysis of the electrolyte, leading to irreversible structural changes and reduced ionic conductivity, as well as the production of toxic H2S gas. Therefore, the synthesis, storage and processing of sulfide electrolytes require strict anhydrous conditions to maintain the stability and safety of the material structure. This obviously increases the complexity and cost of the production process and limits its large-scale production and application. Based on the soft and hard acid-base theory, researchers are currently developing air-stable sulfide electrolytes through oxide blending or element doping.
2.3 Organic/Inorganic Composite Solid Electrolytes
Currently, single inorganic solid electrolytes and polymer solid electrolytes have various problems such as low ionic conductivity, dendrite generation, and unstable interfaces, respectively, and cannot meet the performance requirements of all-solid-state lithium metal batteries. To overcome the shortcomings of inorganic solid electrolytes and polymer electrolytes, inorganic fillers are added to the polymer matrix to form organic/inorganic composite solid electrolytes, it can not only improve the ionic conductivity of polymer solid electrolytes, but also inhibit dendrite generation, improve mechanical strength, improve interface stability and compatibility, etc.
Composite solid electrolytes combine the advantages of inorganic solid electrolytes and organic solid electrolytes. The solid electrolyte obtained by adding inorganic fillers to the polymer solid electrolyte has excellent comprehensive properties, inorganic fillers can play three roles:
- Reduce crystallinity and increase the amorphous phase area, which facilitates Li+ migration;
- Fast Li+ channels can be formed near filler particles;
- Increase the mechanical properties of the polymer matrix, making it easier to form films .
Organic-inorganic composite electrolytes can improve lithium-ion conductivity. There are three main lithium ion conduction mechanisms:
- The organic phase conducts lithium ions;
- The inorganic phase conducts lithium ions;
- The organic-inorganic interface conducts lithium ions.
3. Problems Faced by All solid-state Batteries Research
3.1 Low Ionic Conductivity
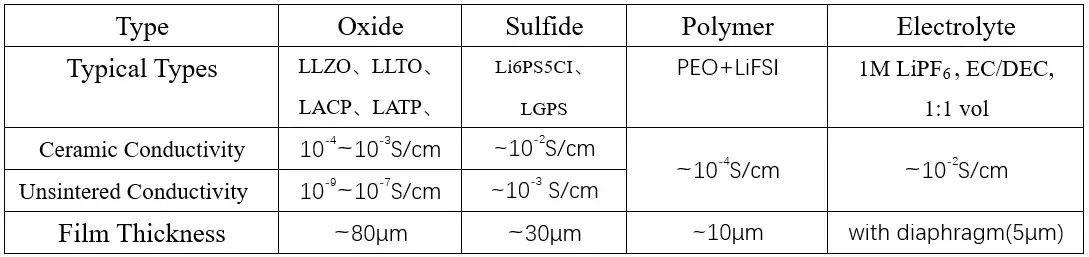
If all liquid electrolytes are replaced with solid electrolytes and all solid-state batteries are constructed, one must first consider whether the lithium-ion conductivity of the solid electrolyte itself is sufficient. The ionic conductivity of the liquid electrolyte is 10-2S/cm, and the ionic conductivity of the oxide solid electrolyte after being sintered into ceramics is 10-4S/cm, the powder state is directly cold pressed to form ~10-8S/cm, and the oxide cannot be directly used in all solid-state batteries. The ionic conductivity of polymer solid electrolyte at room temperature is ~10-6S/cm ~10-5S/cm, and it can only reach ~10-3S/cm when the temperature rises to about 60°C, however, when the temperature rises to 60°C, the polymer solid electrolyte is close to a molten state and is no longer a solid electrolyte in the true sense. Compared with oxide solid electrolytes and polymer solid electrolytes, sulfide solid electrolytes have higher ionic conductivity, the room-temperature ionic conductivity after sintering into ceramics is close to that of liquid electrolytes. From the perspective of conductivity, all solid-state batteries can only place their hope on sulfide all-solid electrolytes and organic/inorganic composite solid electrolytes.
Table 1. Comparison of Three Common Solid Electrolytes and Liquid Electrolytes
3.2 Poor Solid-solid Physical Contact
As shown in Figure 4, in a liquid lithium-ion battery, the flowing organic electrolyte can fully wet the electrode sheet and fill the pores of the electrode, a good liquid-solid infiltration interface is formed, thereby establishing a penetrating and efficient lithium-ion transmission channel from the positive electrode to the negative electrode inside the battery. However, the solid electrolyte in all solid-state batteries cannot flow, which means that the wetting effect of the solid-state contact between the solid electrolyte and the positive and negative electrode particles is far inferior to that of the liquid-solid interface, and the effective contact area is much smaller than that of the liquid electrolyte and the solid particles of the positive and negative electrodes.
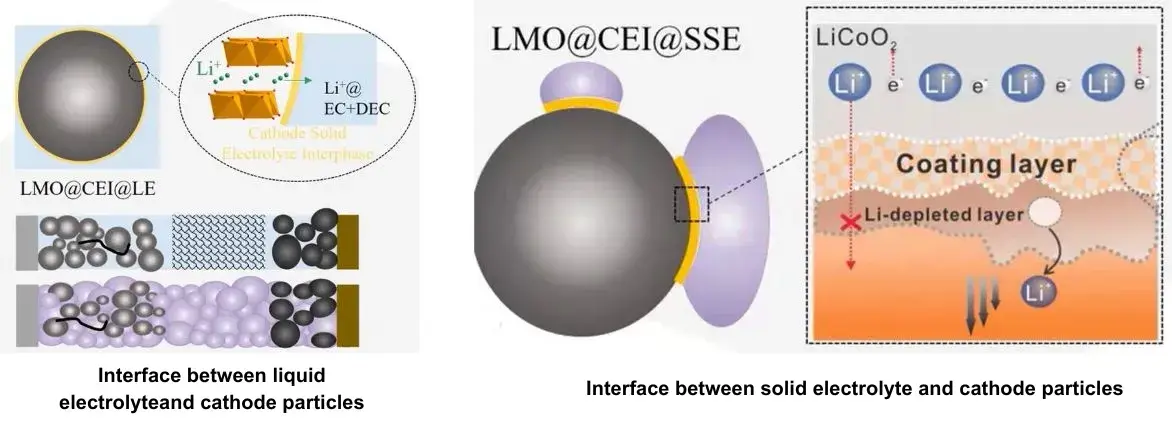
Figure 4. Comparison of the interface between liquid electrolyte and solid electrolyte and cathode particles
To overcome the problems caused by solid-solid physical contact in all solid-state batteries, researchers improved the solid-solid interface by growing solid electrolytes in situ on the electrode materials, this coating layer effectively improves the solid-solid physical contact inside the electrode and reduces the interface impedance. In addition, reducing the porosity inside the electrode through hot or cold pressing processes is also a simple and effective strategy to improve the physical contact between electrode components.
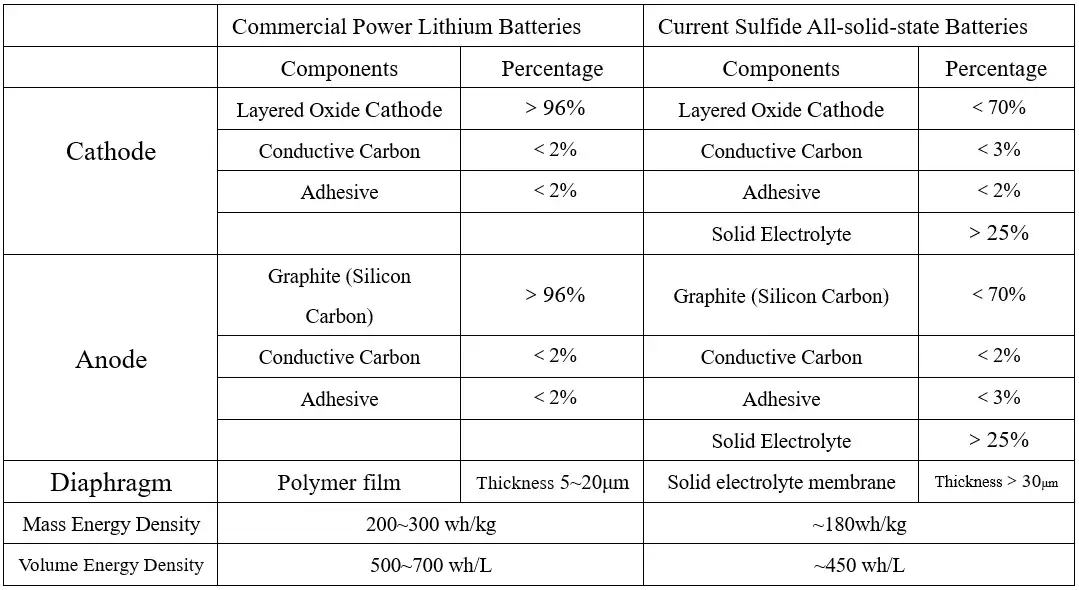
In addition, considering that the lithium-ion conductivity and interfacial infiltration of the solid electrolyte are not as good as those of the liquid electrolyte, it is necessary to increase the volume percentage of the solid electrolyte in the positive and negative active material materials to ensure the lithium-ion conductivity of the positive and negative electrode sheets. Currently, in sulfide all solid-state batteries, more than 25% solid electrolyte needs to be added to the positive and negative electrodes to ensure certain electrical performance. Adding a large amount of solid electrolyte to the electrode reduces the proportion of active material and the mass capacity density of the battery; on the other hand, it increases the volume of the electrode and reduces the volume capacity density of the battery. At the same time, the electronic conductivity of the solid electrolyte itself is very low. Without adding an electronic conductive agent, increasing the volume percentage of the solid electrolyte will lead to a decrease in the electronic conductivity of the electrode, affecting the battery rate performance.
Table 2. Comparison of Parameters Between Commercial Lithium-ion Batteries and Current Sulfide All Solid-state Batteries
3.3 Interface Chemistry/Electrochemical Side Reactions
The ion conduction at the contact interface between solid electrolyte and electrode is mainly affected by the chemical/electrochemical compatibility of the interface, which is mainly reflected in the diffusion of interface elements, interface electrochemical side reactions and space charge layer. The (electro)chemical incompatibility between the sulfide electrolyte and the electrode material causes irreversible parasitic reactions at the interface, forming a complex interface phase with high electronic conductivity and low ion conductivity, further hindering the rapid transport of ions at the interface. In addition, the difference in lithium chemical potential between the sulfide electrolyte and the layered cathode material may lead to the formation of an interface space charge layer and also hinder the migration of lithium ions at the interface.
To improve the interface compatibility between the solid electrolyte and the cathode material and suppress the occurrence of interface (electro)chemical side reactions, the most used and effective method at present is to cover the surface of the cathode material to build an interface protective layer. The cathode material coating in all solid-state batteries is usually a fast ion conductor. These coating materials are required to have high electrochemical stability, high ionic conductivity and electronic insulation, aiming to suppress interface side reactions while not hindering the transport of lithium ions across the interface. Among them, LiNbO3 is favored due to its relatively high ionic conductivity (~10-6 S/cm) and high electrochemical stability with sulfide and cathode materials, and is currently the most widely used coating material.
3.4 Non-uniform Lithium Deposition/Stripping
The uneven lithium deposition/stripping process at the interface between the electrolyte and the metallic lithium anode will lead to the nucleation and growth of lithium dendrites, which may penetrate the electrolyte layer, causing short circuit failure of the battery and posing potential safety hazards. The interfacial contact, interface (electro)chemical stability issues and dendrite growth issues are coupled with each other, making the evolution process of interface dendrites extremely complicated. During the cycle of all solid-state batteries, applying appropriate stacking pressure can effectively improve the physical contact at the interface, and can also promote the creep of lithium and prevent the formation of voids during the peeling process. The shear modulus at the grain boundaries of the electrolyte is about 50% lower than that of its granular phase, and dendrites easily accumulate and propagate along the grain boundaries. Therefore, increasing the density of the solid electrolyte layer and ensuring uniform contact between the electrolyte and the lithium anode interface are effective means to inhibit dendrite growth.
4. Summary of All Solid-state Batteries Research
- The ionic conductivity of oxide and polymer solid electrolytes is too low to be used in all solid-state batteries. We can only rely on sulfide solid electrolytes or organic-inorganic composite electrolytes;
- The commercial application of sulfide solid electrolytes needs to first solve the problems of instability and high cost.
- Replacing electrolyte with solid electrolyte may lead to problems such as deterioration of the electrode plate ion conductive network, poor solid-solid contact, and reduced energy density.
- All solid-state batteries are very difficult to research, but their potential safety and energy density advantages are still worthy of further study.
5. All Solid-state Batteries Test Case
5.1 A comprehensive electrochemical performance testing method for solid electrolytes
The solid electrolyte testing system SEMS1100 (jointly developed by IEST and Xiamen University) is a multi-functional testing system dedicated to solid electrolyte samples. It is a fully automatic measurement equipment for the electrochemical properties of solid electrolytes that integrates tableting, testing and calculation. The system adopts an integrated structural design, including a pressurization module, an electrochemical test module, a density measurement module, a ceramic sheet pressing and clamping module, etc. It is suitable for testing various types of oxides, sulfides, polymers and other solid electrolytes.
Figure 5. Schematic of solid electrolyte testing equipment
5.2 Powder Tablets
At present, when evaluating the electrochemical performance of solid electrolyte powder, it is usually necessary to press the powder into tablets, and samples with poor interface contact also need to spray conductive metal on the surface as an ion blocking electrode. The size and uniformity of the force applied during tableting will greatly affect the integrity of the prepared ceramic tablets. Figure 6 shows macro photos of ceramic tablets obtained by different tableting equipment, among them, the uniform pressure of SEMS1100 equipment is used to prepare solid electrolyte ceramic sheets, which can ensure complete and uniform samples in different pressure ranges, reduce the risk of sample damage, and improve yield and testing efficiency.

Figure 6. Comparison of production results of different equipment
5.3 Ion Conductivity Test
SEMS1100 was used to conduct electrochemical impedance spectroscopy and ionic conductivity tests on two different solid electrolyte materials, Li1.3Al0.3Ti1.7(PO4)3 (LATP) and Li6.5La3Zr1.5Ta0.5O12 (LLZO). As shown in Figure 7, by applying different quantitative pressures to the sandwich ceramic piece and measuring its electrochemical impedance spectrum, it was found that the test pressure will affect its ionic conductivity to varying degrees, this shows that it is necessary to test the electrochemical performance of solid electrolytes by applying stable and quantified pressure.
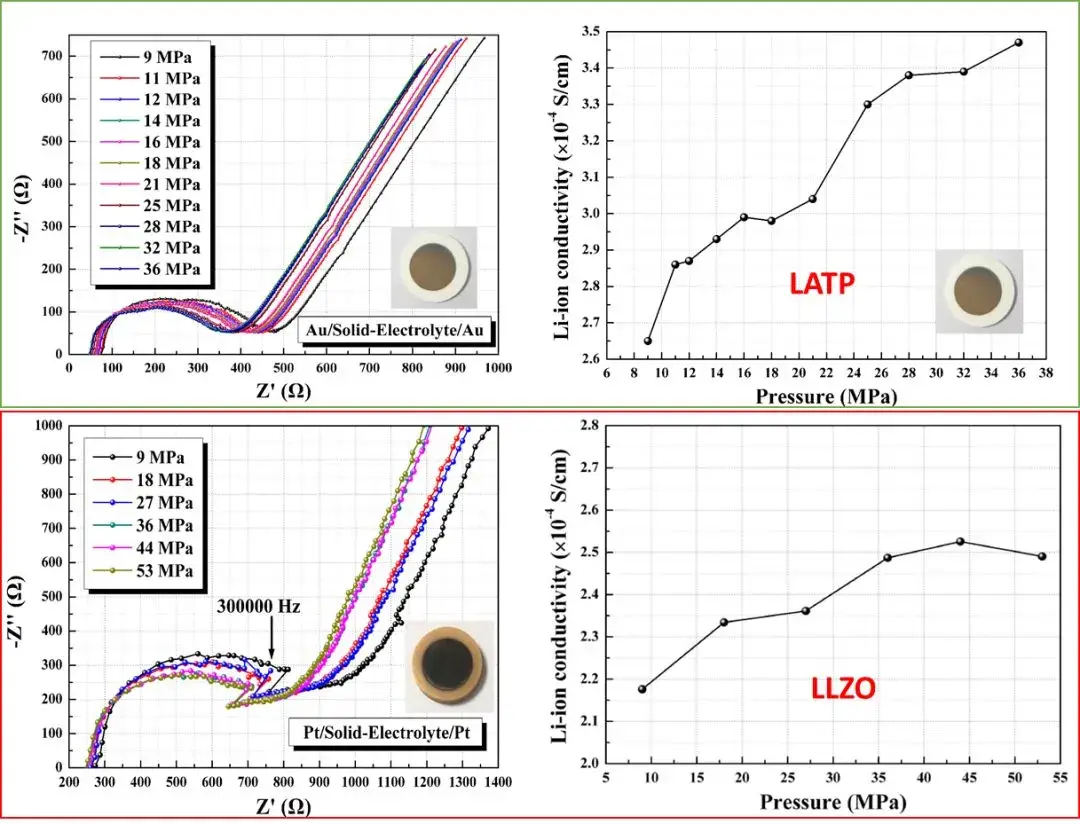
Figure 7. Electrochemical impedance spectra of two solid-state electrolytes and their ionic conductivity changes with pressure.
5.4 Electronic Conductivity & Compaction Density Test
Through the SEMS1100 equipment, the electronic conductivity and compaction density of LATP powder can be simultaneously tested during the tableting process. As shown in Figure 8, as the applied pressure increases, the compacted density increases from the initial 1.7g/cm3 to 2.1g/cm3, the electronic conductivity reaches stability around 50MPa, that is, the density and electronic conductivity trends of the solid electrolyte material are not completely consistent. This shows that under different pressure conditions, different characteristic indicators of solid electrolytes need to be tested simultaneously to obtain comprehensive and accurate measurement results.
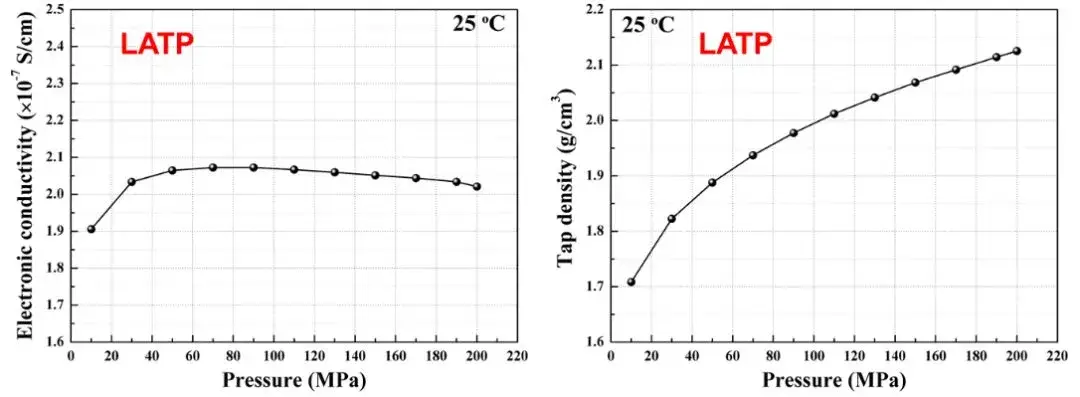
Figure 8. Changes in electronic conductivity and compacted density of LATP solid electrolyte to pressure
5.5 Cycle Performance of Solid-state Lithium Metal Batteries
Assemble the Li-SE-Li symmetrical battery in the sealed fixture provided by SEMS1100, apply different pressures to the battery, conduct cyclic deposition tests of lithium metal, and measure the potential changes of the symmetrical battery. As shown in Figure 9, when the applied pressure is reduced from 120MPa to 110MPa, the overpotential of the battery increases significantly, this shows that the deposition behavior of lithium metal batteries is relatively sensitive to pressure changes, and it is of great significance to evaluate the interfacial stability of solid electrolytes by changing the applied pressure.
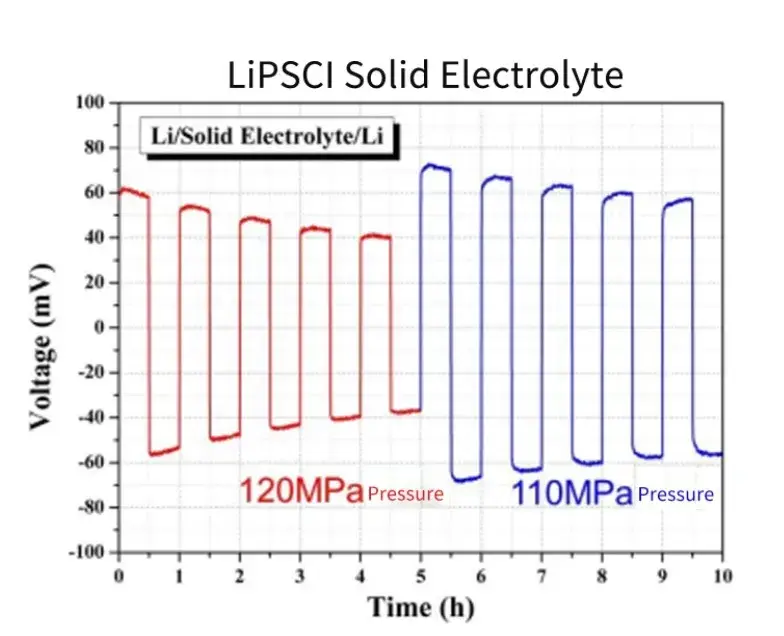
Figure 9. Cyclic charge-discharge test of symmetrical battery
5.6 Electrochemical Stability Window Test
Assemble the Li-SE-stainless steel battery in the sealed fixture provided by SEMS1100 and conduct cyclic voltammetry to test its redox potential. As shown in Figure 10, when the overpotential increases to 3V, the oxidation current density of the battery is only about 1.2μA/cm2, this shows that the solid electrolyte is relatively stable within the voltage window of 0~3V, that is, the SEMS1100 equipment can realize pressurized and sealed electrochemical tests of different solid electrolyte materials and their lithium metal batteries.
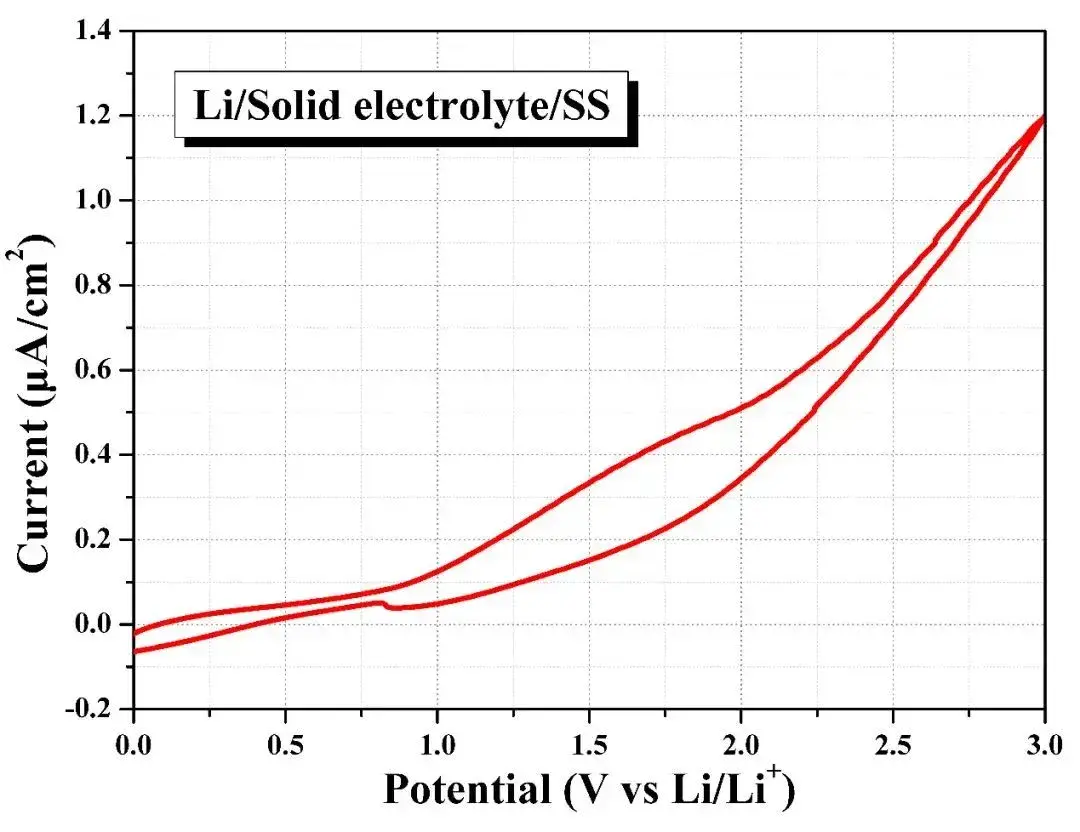
Figure 10. Electrochemical window testing of solid electrolytes
6. References
[1] Liang Yuhao. Research on interface regulation of sulfide-based all solid-state batteries [D]. Beijing: University of Science and Technology Beijing, 2023.
[2] FanL-Z, He H, Nan C-W. Tailoring inorganic-polymer composites for the mass production of solid-state batteries[J]. Nature Review Materials, 2021, 6(11):1003-1019.
[3] Meyer W H. Polymer electrolytes for lithium-ion batteries[J]. Advanced Materials,1998,10(6): 439-448.
[4] Allen J L, Wolfenstine J,Rangasamy E,et al . Effect of substitution (Ta, Al, Ga) on the conductivity of Li7La3Zr2O12[J]. Journal of Power Sources,2012,206(15): 315-319.
[5] Zhu Y, Mo Y. Material design principles for air-stable lithium/sodium solid electrolytes[J]. Angewandte Chemie International Edition,2020,59(40): 17472-17476.
Subscribe Us
Contact Us
If you are interested in our products and want to know more details, please leave a message here, we will reply you as soon as we can.



