-
iestinstrument
Rate-Dependent Failure Behavior Regulation of LiFePO4 Cathode via Functional Interface Engineering
1. Article Information
Title: Rate-Dependent Failure Behavior Regulation of LiFePO4 Cathode via Functional Interface Engineering
First Author: Rui Tang
Corresponding Authors: Jinyang Dong*, Yuefeng Su*, Lai Chen*
Affiliations: Beijing Institute of Technology; Beijing Institute of Technology Chongqing Innovation Center; China Electric Power Research Institute; State Grid Henan Electric Power Research Institute
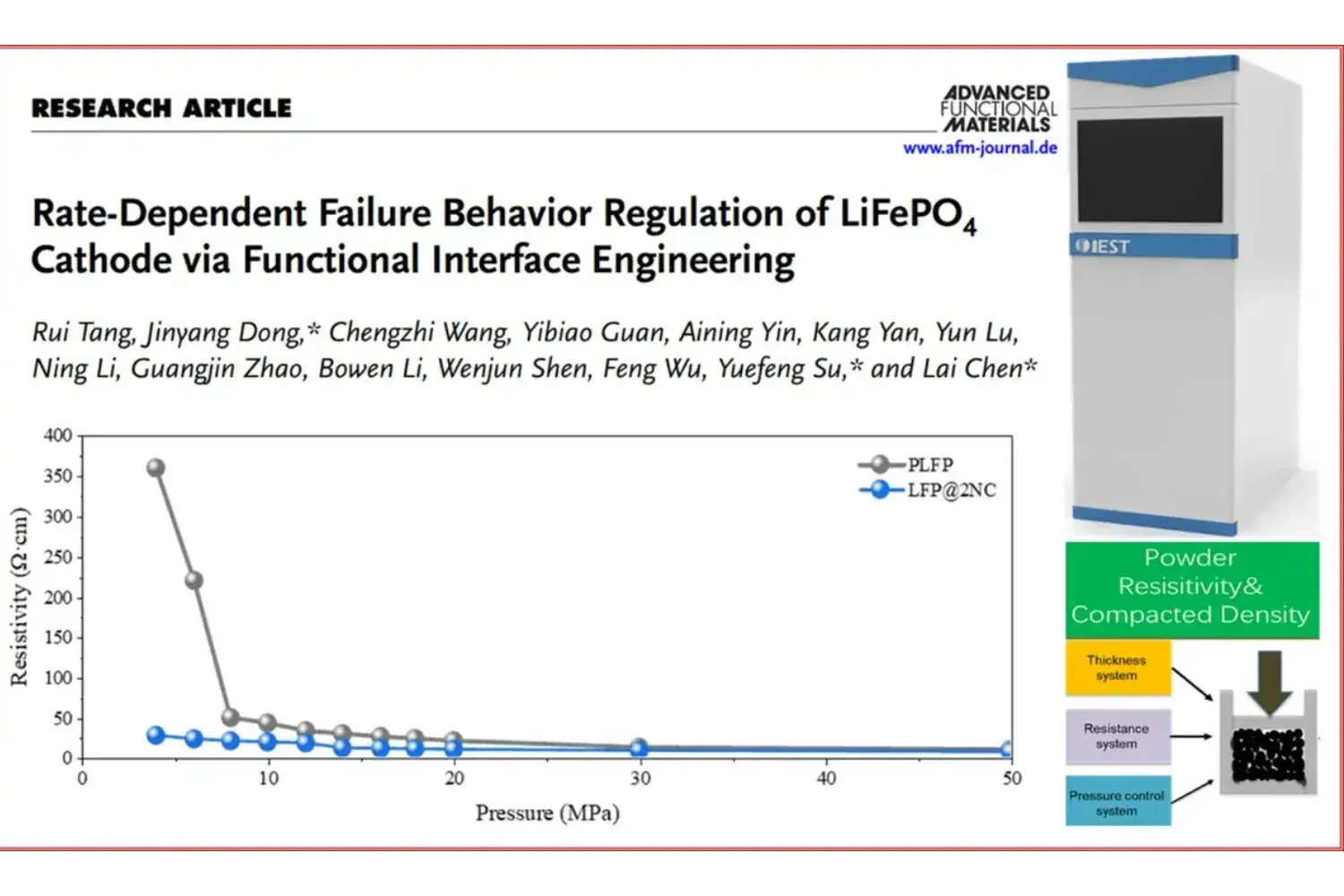
Instrumentation: IEST Powder Resistivity & Compaction Density Measurement System (PRCD3100)
2. Research Background
The demand for high-performance lithium-ion batteries is driven by sustainable economic development and shifts in energy structure. LiFePO₄ batteries have attracted extensive attention due to their excellent thermal stability and low cost. However, the intrinsic low electronic conductivity and limited lithium-ion diffusion coefficient of LiFePO₄ restrict its further application under high-rate conditions. Although many established strategies—such as coating, doping, and morphology control—have been employed to improve the kinetic properties and stability of the material, the intrinsic relationship between kinetics and stability remains insufficiently explored. As the charge/discharge rate directly relates to the power requirements of battery users, understanding the differences in failure behavior between low-rate and high-rate cycling is critical to advancing the application of LiFePO₄ in fast-charging/discharging scenarios.
The primary cause of LiFePO₄ degradation is the irreversible loss of active lithium; that is, lithium released during charging cannot be fully reinserted into the lattice during discharging, leading to capacity fade. At higher cycling rates, increased stress, strain, and impedance facilitate irreversible phase transitions. Furthermore, the inherently sluggish kinetics of LiFePO₄ lead to non-uniform lithium extraction/insertion, exacerbating structural degradation under high-rate conditions. Conversely, at lower cycling rates, reduced polarization allows for more complete lithiation/delithiation; however, the extended reaction time results in the accumulation of side products and consumption of active lithium, thereby affecting cycle life. These observations indicate that the failure mechanisms differ substantially with cycling rate, necessitating tailored improvement strategies for each failure mode.
3. Overview of the Work
Recently, Professor Yuefeng Su, Researcher Lai Chen, Postdoctoral Researcher Jinyang Dong, and their colleagues at Beijing Institute of Technology have successfully regulated the rate‐dependent failure behavior of LiFePO₄ through the design of functional interface engineering. Under high charge/discharge rates, the functional interface layer, by exploiting the affinity between nitrogen and lithium, promotes lithium-ion migration, reduces internal polarization, and alleviates mechanical stress, thereby suppressing structural degradation. Under lower rates, the functional interface layer contributes to the formation of a stable cathode–electrolyte interphase (CEI), effectively inhibiting side reactions and minimizing active lithium loss. Through a combined experimental and theoretical approach, the key mechanisms of rate‐dependent failure regulation have been elucidated, advancing the potential application of LiFePO₄ in advanced energy storage systems requiring rapid charge/discharge and long-term cycling stability. This work is published in the top-tier international journal Advanced Functional Materials under the title “Rate‐Dependent Failure Behavior Regulation of LiFePO₄ Cathode via Functional Interface Engineering.”
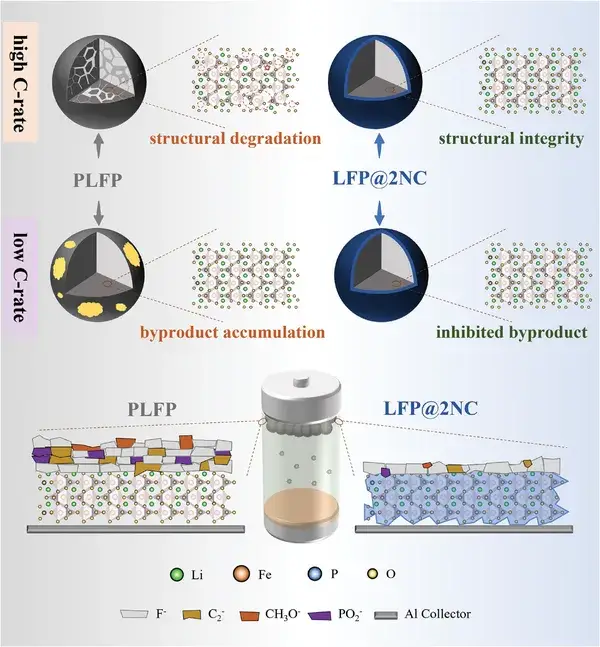
Figure 1. Schematic diagram of failure mechanism at different cycle rates
4. Key Points of the Article
4.1 Verification of Rate‐Dependent Failure Behavior Regulation via Functional Interface Engineering
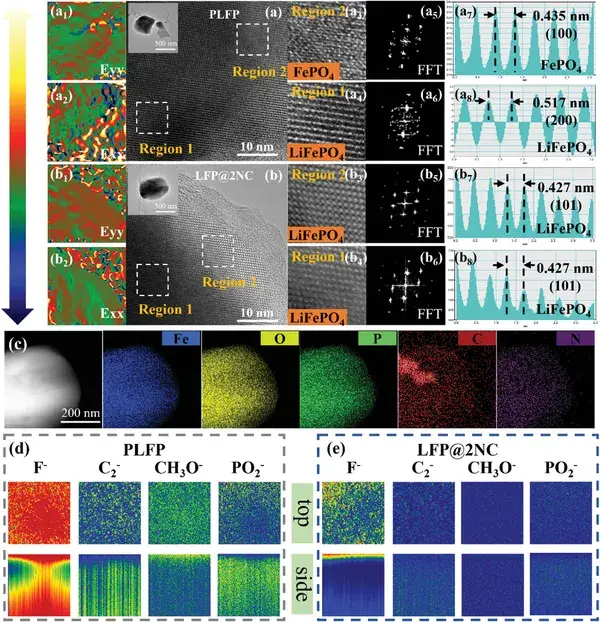
Figure 2. Analysis of the cycled structure and surface composition.
High-resolution transmission electron microscopy (HRTEM) and time-of-flight secondary ion mass spectrometry (TOFSIMS) were conducted on cycled samples to analyze changes in structure and surface composition. HRTEM images reveal that the PLFP sample exhibits two distinct phases—FePO₄ and LiFePO₄—whereas the LFP@2NC sample retains a single-phase LiFePO₄ structure, indicating that the PLFP sample has undergone an irreversible phase transition. Moreover, geometrical phase analysis (GPA) shows that the interface between the two phases in the PLFP sample exhibits higher tensile stress, which may be the root cause of the irreversible phase transition. TOFSIMS results further demonstrate differences in surface composition, with the LFP@2NC sample showing markedly less accumulation of side reaction products. Thus, the functional interface layer regulates the rate-dependent failure behavior of LiFePO₄ through both the reduction of stress/strain and the suppression of side product deposition.
4.2 Impact of Functional Interface Engineering on Initial Charge/Discharge Behavior

Figure 3. Evolution of surface composition and structural changes during the first week of charge/discharge.
Investigations were performed to elucidate the modification mechanism of functional interface engineering during the initial week of charge/discharge. Non-in-situ X-ray photoelectron spectroscopy (XPS), in-situ electrochemical impedance spectroscopy (EIS) with distribution of relaxation times (DRT) analysis, and in-situ X-ray diffraction (XRD) were carried out on both PLFP and LFP@2NC samples. Non-in-situ XPS results reveal that the LFP@2NC sample does not exhibit a LiF signal throughout the first week, whereas in the PLFP sample a LiF signal appears at the end of charging and disappears at the end of discharging, indicating a tendency for repeated consumption of active lithium and accumulation of surface side products. In-situ EIS and in-situ XRD results both indicate that the interface-modified material exhibits improved cycling reversibility under identical charge/discharge conditions, signifying enhanced kinetic properties.
4.3 Theoretical Calculations and Simulations on the Regulation Mechanism
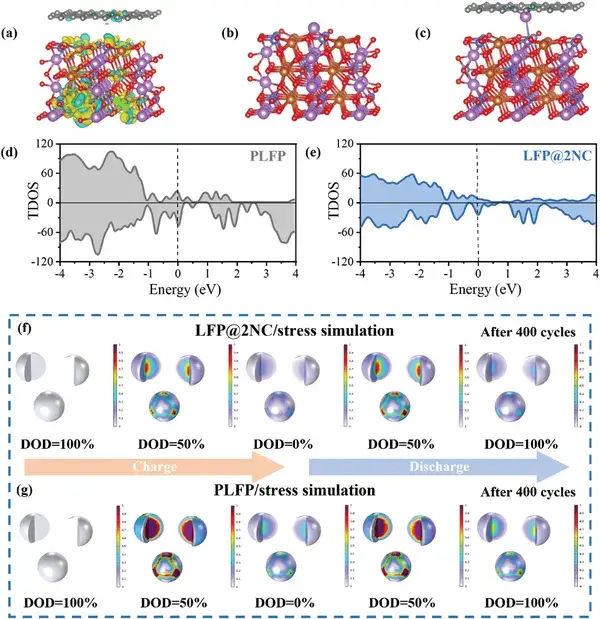
Figure 4. Results from theoretical calculations and simulations.
To further elucidate the mechanism by which functional interface engineering regulates the rate-dependent failure behavior, theoretical calculations and simulations were performed. Differential charge density analysis reveals that charge transfer occurs between Fe and N within the interface layer, which may strengthen the Fe–O bonds, thereby conferring enhanced structural stability to LiFePO₄ under high-rate cycling conditions. Adsorption energy calculations confirm the strong affinity between nitrogen and lithium, laying the foundation for improved lithium-ion migration. Additionally, simulation results substantiate the stress-relieving effect of the functional interface engineering during charge/discharge cycles. The combined effects of enhanced lithium-ion migration, improved structural stability, and reduced stress accumulation endow the functional interface with the capability to regulate the failure behavior of LiFePO₄ across different rates.
5. Conclusions
This work demonstrates that functional interface engineering can effectively regulate the rate‐dependent failure behavior of LiFePO₄. Under high-rate cycling, the failure of LiFePO₄ is primarily driven by structural degradation induced by stress accumulation, whereas under low-rate cycling, the failure is mainly due to the accumulation of side reaction products and the consumption of active lithium. The nitrogen-based interface layer is designed to enhance lithium-ion migration kinetics, reduce stress, and prevent structural degradation under high-rate conditions, while also participating in the formation of a stable CEI to inhibit the accumulation of side reaction products and the loss of active lithium under low-rate conditions. This dual regulation mechanism has been successfully validated in both button-type half-cells and Ah-level soft-pack full cells, offering a potential solution for high-power energy storage applications.
6. Reference
Rui Tang, Jinyang Dong*, Chengzhi Wang, Yibiao Guan, Aining Yin, Kang Yan, Yun Lu, Ning Li, Guangjin Zhao, Bowen Li, Wenjun Shen, Feng Wu, Yuefeng Su*, and Lai Chen*. Rate‐Dependent Failure Behavior Regulation of LiFePO₄ Cathode via Functional Interface Engineering. Advanced Functional Materials, 2025: 2421284.
https://doi.org/10.1002/adfm.202421284
7. Related Testing Instrument Recommendations: IEST Powder Resistivity & Compaction Density Tester (PRCD3100)
Introduction: PRCD3100 combined with high-precision pressure control,thickness and resistance testing system, free choice of four-probe and two-probe method. Accurately test the resistivity and compaction density of powders for material research and development and batch stability monitoring of powder materials.
Subscribe Us
Contact Us
If you are interested in our products and want to know more details, please leave a message here, we will reply you as soon as we can.