-
iestinstrument
Entering Electrochemistry | Revealing the Inner Workings of Lithium-Ion Cells Through EIS Testing
1. Introduction
Electrochemical Impedance Spectroscopy (EIS) functions like a non-destructive X-ray for batteries: it decomposes the cell’s internal electrochemical and transport processes across different time scales. In this report we compare the performance of IEST ERT7008 (ERT7 series) against a well-known foreign electrochemical workstation to evaluate how closely their EIS measurements agree in common cell formats and test modes. The goal is to present the methods, key spectral interpretations, and practical implications for battery R&D teams performing EIS testing and dynamic EIS measurements.
2. Fundamentals of EIS Testing
EIS, or Electrochemical Impedance Spectroscopy, characterizes a battery’s AC impedance across a frequency spectrum by applying a small-amplitude sinusoidal signal. Data is typically presented as a Nyquist plot (Z’ vs Z”). This technique non-invasively probes the impedance characteristics of various interfaces and bulk phases within a cell, including electron/ion conduction, interfacial reaction kinetics, and mass transport processes. By analyzing the EIS spectrum’s response in different frequency regions, key parameters such as SEI film stability, charge transfer efficiency, and lithium-ion diffusion rates can be quantitatively assessed. This provides crucial data for optimizing battery materials, diagnosing aging mechanisms, and enhancing electrochemical performance.
2. Applications of EIS in Lithium-Ion Battery Research
Understanding the Li⁺ transport pathway within a cell helps correlate specific processes with features on the Nyquist plot, enabling the deduction of internal electrochemical mechanisms from the obtained curves.
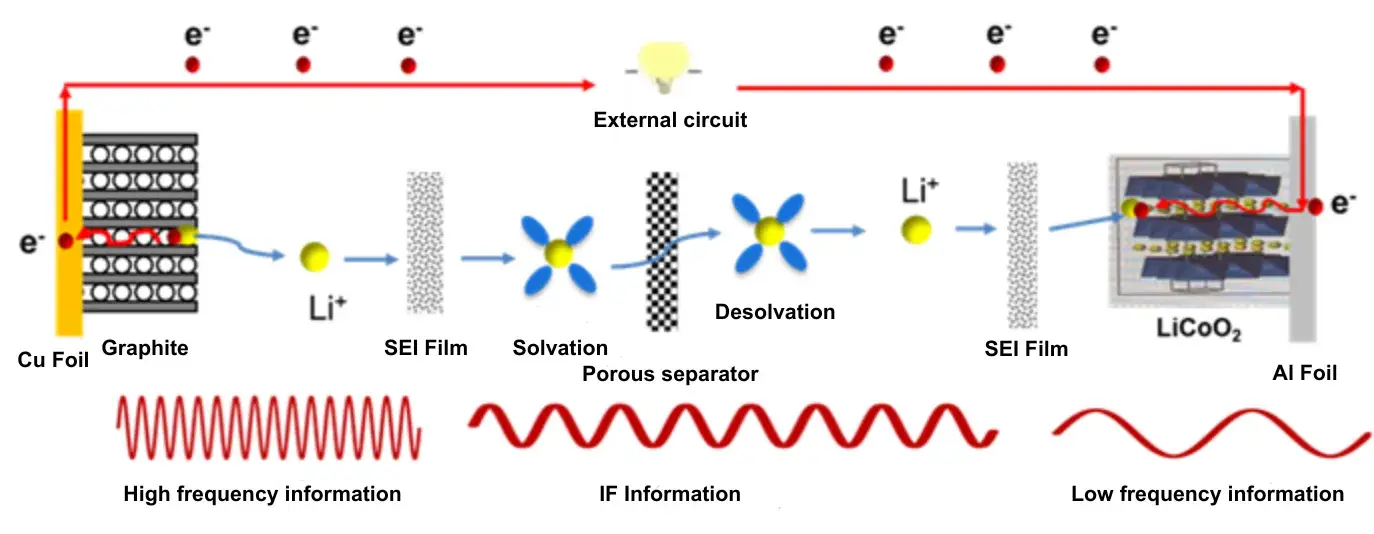
Figure 1. Li⁺ transport pathway in a lithium-ion battery.
2.1 The diagram illustrates the Li⁺ transport sequence:
The sequence is as follows:
-
External circuit: Electron conduction (Cu current collector → Graphite anode).
-
Graphite anode: Li⁺ intercalation into graphite, passing through the SEI film.
-
Electrolyte: Li⁺ solvation and migration through the porous separator.
-
Cathode (e.g., LiCoO₂): Li⁺ de-solvation, potential passage through a surface layer, and intercalation into the cathode material.
-
External circuit: Electron conduction via the Al current collector.
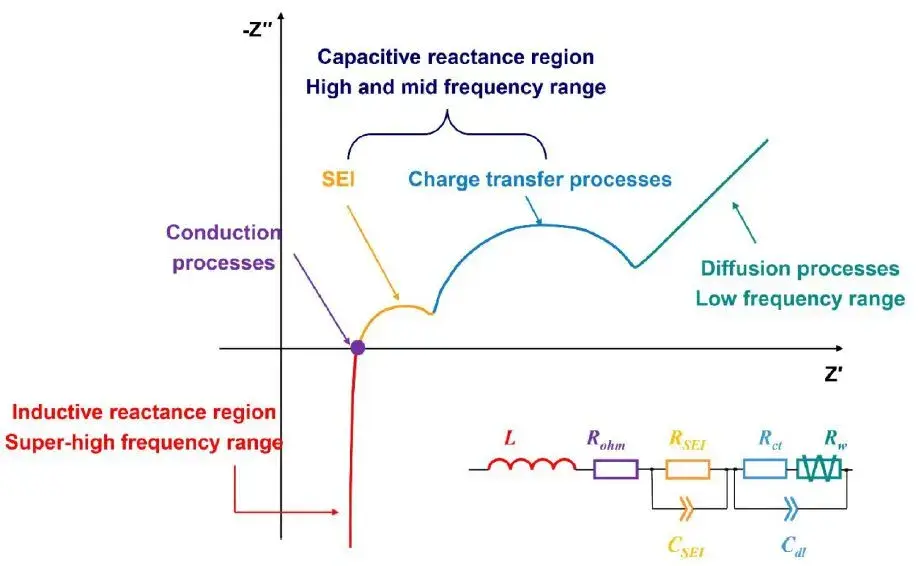
Figure 2. Typical EIS Nyquist plot for a Li-ion battery with characteristic frequency regions identified.
The high-frequency region reflects the ionic transport characteristics across the SEI layer, the mid-frequency region corresponds to the charge transfer reaction, while the low-frequency region characterizes solid-state diffusion kinetics within the electrode material. By deconvoluting the impedance response across distinct EIS frequency domains, key electrochemical parameters governing interfacial and bulk processes can be quantitatively evaluated, including SEI film resistance (R_SEI), charge transfer resistance (R_ct), and diffusion impedance (R_w).
2.2 High-Frequency Region (First Semicircle)
- Corresponding Process: Ionic transport impedance of Li⁺ through the SEI layer.
-
Electrochemical Parameters:
-
R_SEI: Resistance of the SEI layer.
-
C_SEI: Capacitance of the SEI layer (reflecting its double-layer characteristics).
-
2.3 Mid-Frequency Region (Second Semicircle)
- Corresponding Process: Charge transfer reaction (electrochemical reaction) at the electrode/electrolyte interface, including:
-
Anode: Li⁺ intercalation/deintercalation in graphite.
-
Cathode: Li⁺ intercalation/deintercalation in LiCoO₂.
-
Electrochemical Parameters:
-
R_ct: Charge transfer resistance (indicates kinetics of electrochemical reactions).
-
C_dl: Double-layer capacitance (charge accumulation at the electrode/electrolyte interface).
-
2.4 Low-Frequency Region (Sloping Line)
- Corresponding Process: Solid-state diffusion of Li⁺ within the bulk electrode material (e.g., inside graphite or LiCoO₂ particles).
-
Electrochemical Parameters:
-
R_w: Warburg impedance (diffusion impedance, inversely proportional to the square root of frequency).
-
3. Experimental Comparison and Results
3.1 Experimental Setup:
Two distinct cell configurations were selected for EIS testing:
- Cell Comparison: Coin cell vs. Pouch cell
- Equipment Comparison: International electrochemical workstation vs. IEST ERT7008
Coin Cell (4.15V, 25°C), Test Parameters: 100 kHz ~ 0.1 Hz, 10 mV sinusoidal signal amplitude (PEIS).
Pouch Cell (2Ah, 3.19V, 25°C), Test Parameters: 100 kHz ~ 0.1 Hz, 80 mA AC current amplitude (GEIS).
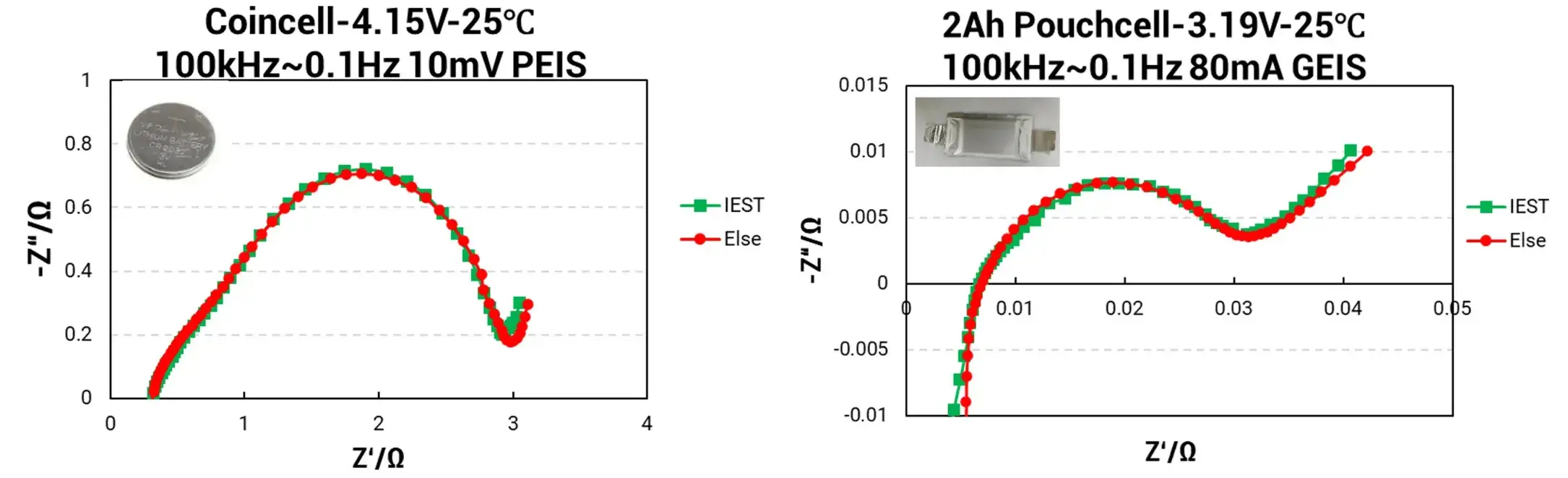
Figure 3. EIS Analysis–Different Cell Impedance Analysis
3.2 Results
The EIS data for the Coin Cell demonstrates exceptional congruence between the IEST ERT7008 (green curve) and the competitor equipment (red curve), indicating a very high degree of impedance data matching. Similarly, EIS results for the Pouch Cell show only minor discrepancies between the IEST system and the international electrochemical workstation, with deviations consistently below 5%.
4. IEST ERT 7 Series Specifications
4.1 Wide Frequency Range: 0.01 Hz ~ 100 kHz
- Frequency range is a critical EIS specification. The low-frequency segment (<1 Hz) analyzes diffusion processes, while the high-frequency segment (>10 kHz) reflects contact resistance. The IEST system covers the broad range of 0.01 Hz to 100 kHz, suitable for diverse cell types.
4.2 High-Precision Signal Control: Accommodates cells from 10 mΩ to kΩ range
-
The instrument accommodates a wide impedance range from milli-ohms (10 mΩ) to kilo-ohms (kΩ), utilizing a high-precision signal source and low-noise measurement system to accurately capture subtle changes.
4.3 Support for Multiple EIS Modes: PEIS & GEIS
- Potentiostatic EIS (PEIS) is suitable for micro-current systems like coin cells and lab-scale cells. Galvanostatic EIS (GEIS) is better suited for pouch cells undergoing low-current discharge. The IEST system supports both modes, catering to varied application requirements.
4.4 Why Choose IEST Instruments Solutions?
| Comparison Item | Electrochemical Workstation | IEST Instruments Solution |
|---|---|---|
| Price | 200,000+ RMB | 50% Cost Reduction |
| Technical Support | Email Communication | 24h Local Technical Support |
| Functional Scope | Stand-alone EIS Testing | Integrated Charge-Discharge & EIS Testing, Supports Dynamic EIS |
-
High Cost-Effectiveness: IEST delivers high-performance testing capabilities at a significantly more accessible price point, eliminating the need for costly additional equipment.
-
Localized Support: Compared to the support response times of international brands, IEST provides faster technical support and after-sales service within the domestic market, ensuring prompt resolution of customer issues.
-
High Integration: The IEST ERT7 series is not merely an EIS tester; it integrates seamlessly with high-precision battery cyclers, enabling comprehensive battery testing and analysis.
5. Featured Application Cases
5.1 EIS Testing at Different SOC and DOD States
Performing impedance tests at various States of Charge (SOC) and Depth of Discharge (DOD) provides a comprehensive view of internal changes during operation. Variations in the EIS curves reveal details about Li⁺ migration, charge exchange rates, and the state of interface films, aiding in performance assessment and degradation trend analysis. This methodology is vital for cell optimization design and lifetime prediction.
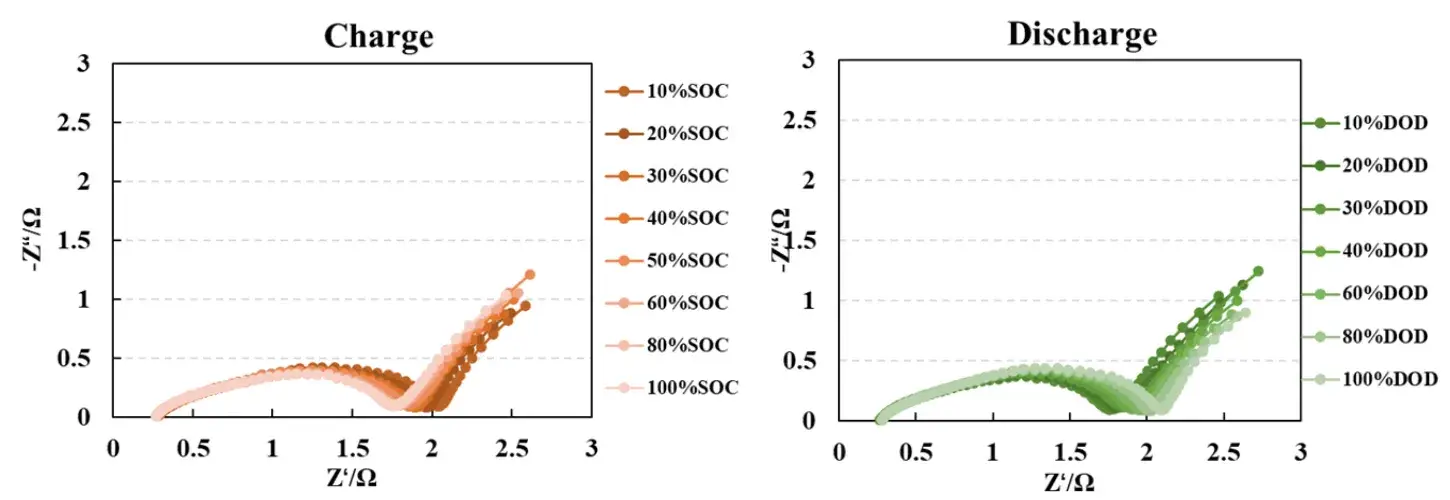
Figure 4. EIS Testing at different States of Charge (SOC) and Depths of Discharge (DOD)
5.2 Dynamic EIS Testing
Dynamic EIS is an advanced technique for tracking internal impedance changes in real-time during battery operation (charging/discharging). It eliminates the need for test interruptions required by traditional methods. This capability allows for observing the battery’s “state of health” across different SOCs, revealing details of electrochemical processes such as Li⁺ transport, charge transfer kinetics, and interface stability. This technology is crucial for battery design optimization, aging analysis, and operational state monitoring. The IEST ERT7 series supports dynamic EIS testing with a DC bias current range from microamps to over 10 Amps, meeting diverse application needs.

Figure 5. Dynamic EIS Testing
7. Conclusion
Our experimental results validate the EIS testing capability of IEST instrument. The ERT7008 demonstrates broad frequency coverage, adequate dynamic range, and continuous dynamic EIS capabilities suitable for both coin and pouch cells. The data shows excellent agreement with a leading international electrochemical workstation, with deviations within 5%, fully meeting the requirements for battery R&D and testing. Coupled with its high cost-effectiveness and responsive localized support, the IEST ERT series presents a compelling alternative for engineers and researchers seeking accurate, efficient, and stable EIS testing solutions.
Contact Us
If you are interested in our products and want to know more details, please leave a message here, we will reply you as soon as we can.



