-
iestinstrument
IEST Facilitates High-Nickel Cathodes Development for Stability and High-Rate Performance
1. Abstract
High-nickel cathode materials offer high capacity and favorable cost-per-energy, but their adoption at high voltage and fast charge rates is hampered by structural instability and stress accumulation. This paper presents a metal-cation-induced “three-in-one” surface modification that combines W-doping, a reconstructed cation-mixed surface layer, and a thin Li₂WO₄ nano-coating to simultaneously strengthen the surface oxygen bonding, accelerate Li⁺ transport, and suppress side reactions. The approach improves chemo-mechanical stability and high-rate cycling, and clarifies the rate-failure mechanisms through combined macroscopic and single-particle characterization.
2. Introduction
High-nickel cathodes are increasingly attractive for high-energy lithium-ion batteries because they deliver high specific capacity at relatively low cost. However, when cycled to high cutoff voltages or under fast charging, these materials suffer structural degradation, oxygen release, and stress-driven particle fracture that limit cycle life and rate capability. To address these problems, Wu Feng’s group at Beijing Institute of Technology developed a metal-cation-induced three-in-one modification to concurrently enhance surface chemistry, mechanical integrity, and Li⁺ transport in high-nickel materials. The full study entitled “Enhancing Chemomechanical Stability and High-Rate Enhancing Chemomechanical Stability and High-Rate Performance of Nickel-Rich Cathodes for Lithium-Ion Batteries through Three-in-One Modification”is reported in Energy Storage Materials (DOI: 10.1016/j.ensm.2024.103893).
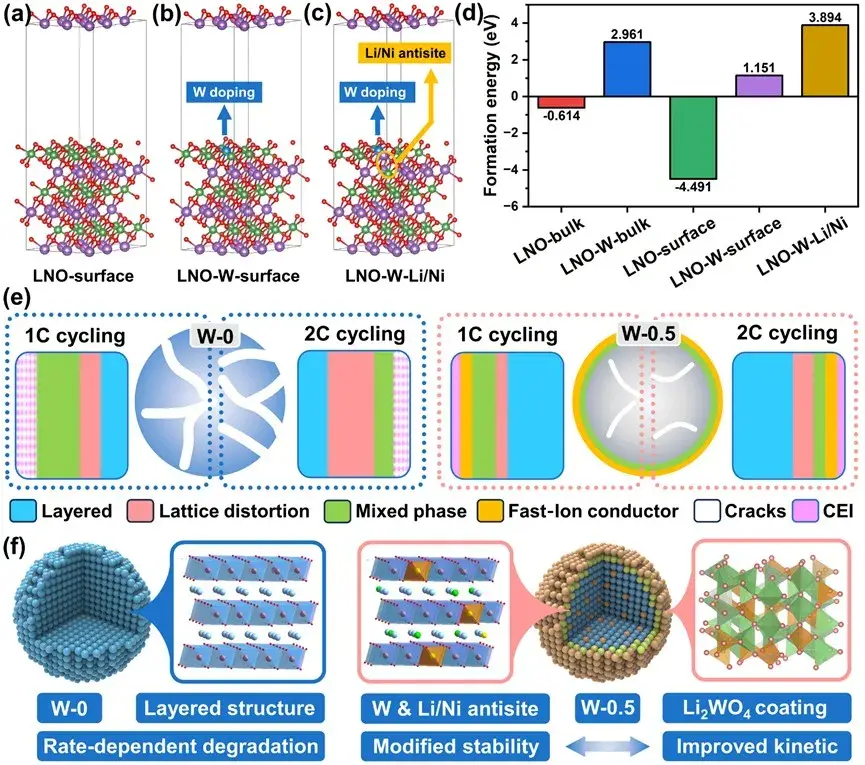
Figure. 1. Schematic diagram of hexavalent metal cation induced three-in-one modification mechanism and multiplicative failure mechanism
3. Three-in-One Modification Strategy
The three-in-one scheme integrates three cooperative modifications at the particle surface:
-
W doping of the surface lattice — Tungsten incorporation strengthens the binding between surface oxygen and the transition-metal sublattice, which reduces oxygen loss during deep delithiation and inhibits the layered→rocksalt phase transition.
-
Reconstructed cation-mixed surface layer — A controlled cation mixing layer with optimized thickness accelerates Li⁺ diffusion near the surface and mitigates structural degradation that begins at the particle exterior.
-
Li₂WO₄ nano-coating — A thin Li₂WO₄ shell reduces parasitic reactions between active material and electrolyte, limiting surface corrosion and gas evolution.
Together, these modifications work synergistically to improve both chemical and mechanical robustness of the particles.
4. Key Experimental Characterization (methods and instruments)
To directly link particle-level properties to electrode performance, the authors combined bulk and single-particle measurements:
-
Powder resistivity / conductivity and tap/packing density were measured using the IEST Powder Resistivity & Compaction Density Tester (PRCD3100). Results show that surface Li₂WO₄ formation consumes residual alkali and lowers powder resistivity, thus increasing bulk electronic conductivity.
-
Single-particle mechanical testing (SPFT2000, IEST) employed a high-precision load cell and displacement control to quantify compressive strength and fracture behavior of individual cathode particles. The three-in-one layer increased compressive strength and reduced displacement at fracture, indicating improved resistance to internal stress and a higher tolerance for compaction.
-
PITT (potentiostatic intermittent titration technique) to extract Li⁺ diffusion coefficients and demonstrate enhanced Li⁺ transport through the reconstructed surface layer.
-
In-situ structural and gas analyses: in-situ XRD, in-situ Raman, and in-situ DEMS were used to probe phase evolution, transition-metal migration, and oxygen/gas release during high-voltage cycling.
-
DFT modeling supplied atomic-scale insight into oxygen vacancy formation energies and transition-metal migration barriers for surfaces with and without the high-entropy (cation-mixed) layer.
Figure 2. Appearance of IEST Powder Resistivity & Compaction Density Tester(PRCD)
Figure 3. (a) IEST Single-Particle Mechanical Properties Test System (SPFT); (b) Test Mode with Controlled Displacement; (c) Bottom View of the Optical System
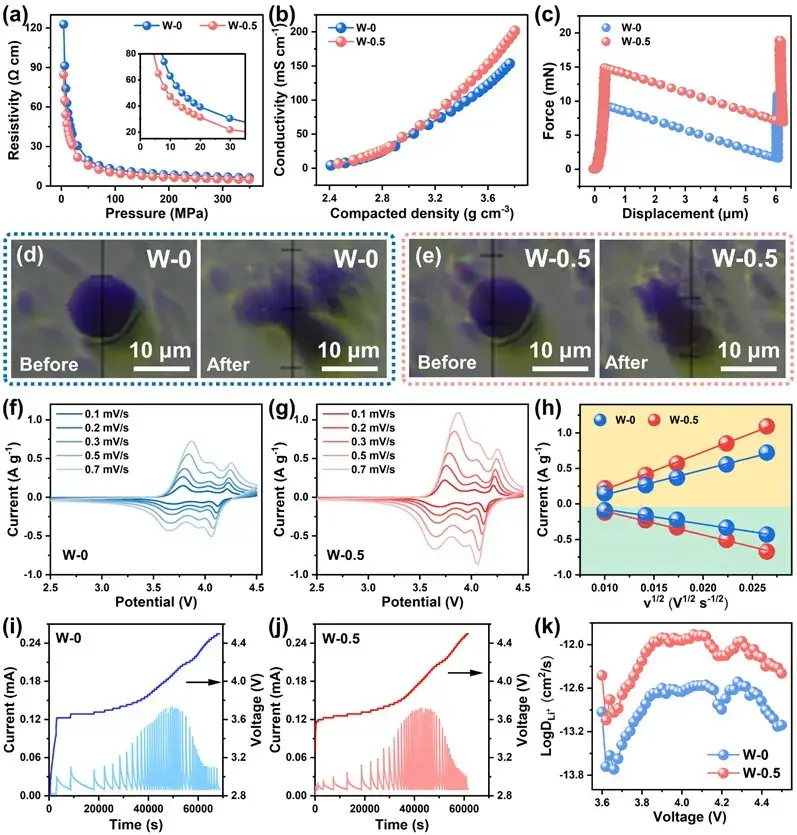
Figure 4. (a) Powder resistivity of W-0 and W-0.5 samples; (b) Electronic conductivity; (c) Single particle force test; (d) Optical photographs of W-0; (e) W-0.5 samples before and after single particle mechanical testing; (f) CV curves of W-0 and; (g) W-0.5 at 0.1-0.7 mV s-1; (h) Relationship between scan rate and peak current; (i) PITT test of W-0; (j) PITT test of W-0.5; (k) Corresponding Li+ diffusion coefficient
5. Single-Particle Mechanical and Electronic Effects
Single-particle compression data reveal that the modified particles sustain larger internal stress before fracturing and exhibit smaller fracture displacements relative to unmodified (W-0) samples. Reduced powder resistivity and higher electronic conductivity correlate with the Li₂WO₄ coating consuming residual surface basic species and improving particle–particle contact after compaction. PITT analysis shows the reconstructed cation-mixed layer combined with the Li₂WO₄ shell yields substantially higher Li⁺ transport efficiency, which directly translates into improved capacity retention at high current densities. These particle-level improvements therefore rationalize the observed gains in electrode-level volumetric capacity and rate capability.
6. Mechanistic Insights into Rate-Failure and Stability
To determine why rate performance degrades in high-nickel cathode materials and how the three-in-one modification mitigates this, the authors systematically compared pre- and post-cycling samples under multiple charge/discharge rates:
-
In-situ XRD: Modified materials show slower changes in lattice parameters at high cutoff voltages, indicating reduced structural strain accumulation and more stable cell volume behavior during delithiation.
-
In-situ Raman: Characteristic peaks near 500 cm⁻¹ and 618 cm⁻¹ remain largely unchanged in modified samples, whereas the unmodified material develops a new peak near 650 cm⁻¹ consistent with formation of a spinel-like phase resulting from transition-metal migration.
-
In-situ DEMS: Gas evolution (oxygen-release related signals) is significantly suppressed for samples with the high-entropy surface layer and Li₂WO₄ coating, indicating a more robust lattice oxygen framework under high-voltage conditions.
-
DFT modeling: Theoretical calculations show higher oxygen vacancy formation energies and larger migration barriers for Mn (and other TM) in the high-entropy surface environment, supporting the experimental observation that transition-metal migration and oxygen loss are kinetically disfavored after modification.
Together, these results point to a combined chemical and mechanical stabilization mechanism: strengthened oxygen bonding plus constrained TM migration reduce phase transitions and gas release, while improved surface Li⁺ transport and mechanical integrity preserve particle morphology during fast cycling.

Figure 5. (a) SEM cross-sectional image of W-0 at 1 C; (b) SEM cross-sectional image of W-0.5 after 100 cycles at 1C; (c) W-0 at 2C; (d) SEM cross-sectional image of W-0.5 after 100 cycles at 2 C; (e) W-0 at 1C (f) W-0.5 at 1C; (g) W-0 at 2C; (h) SEM cross-sectional image of W-0.5 after 100 cycles at 2C; (i) W-0 at 1C; (j) W-0.5 at 1 C; (k) W-0 at 2C; (l) GPA analysis of W-0.5 at 2C
7. Long-Term Cycling and Morphological Evidence
SEM cross-sections and GPA (geometric phase analysis) of electrodes after 100 cycles at 1 C and 2 C show that the modified material (W-0.5) preserves particle integrity and reduces internal cracking compared with the unmodified baseline (W-0). The GPA maps indicate smaller local strain accumulation in modified particles across both moderate (1 C) and high (2 C) rates, consistent with the single-particle and in-situ structural analyses. These morphological observations directly support improved capacity retention and rate performance for the three-in-one modified high-nickel cathodes.
8. Summary
This study presents metal-cation-induced three-in-one surface modification offers a generalizable route to improve both chemomechanical stability and high-rate performance of high-nickel cathodes. By systematically combining powder resistivity measurements with single-particle mechanical testing, the research establishes clear correlations between intrinsic material properties and macroscopic cell performance. The findings offer valuable mechanistic insights into high-rate failure mechanisms, providing a scientific foundation for designing more robust and high-performance cathode materials for the next generation of lithium-ion batteries.
9. Original Article
Contact Us
If you are interested in our products and want to know more details, please leave a message here, we will reply you as soon as we can.