-
iestinstrument
The Main Factors Affecting The Compaction Density of Cathode Electrode Sheet
1. Abstract
Compaction density is a primary design parameter for cathode electrode sheets; it strongly influences areal capacity, internal resistance, electrolyte wetting, and cycle life. This article translates and reorganizes experimental findings that identify the dominant factors controlling cathode compaction density—material true density, particle morphology, particle size distribution, and electrode manufacturing process—and offers practical strategies to optimize electrode density for high-performance lithium-ion cells.
2. Introduction
During lithium-ion battery manufacturing, compaction density significantly impacts overall cell performance. This parameter shows strong correlations with electrode specific capacity, efficiency, internal resistance, and cycle life. As a result, identifying the optimal compaction density is essential in cathode powder battery testing and electrode design.
Generally, within a material’s allowable compaction range, higher electrode density enables greater battery capacity, making compaction density a key indicator for estimating material energy density. However, excessively pursuing high density can actually reduce specific capacity and impair cycle performance.
As density increases, particles experience greater compression, reducing electrode porosity. This diminishes electrolyte absorption and wetting capability. The direct consequences include poor specific capacity utilization, reduced electrolyte retention, increased polarization during cycling, accelerated capacity fade, and noticeably higher internal resistance.
Therefore, appropriate cathode compaction density increases discharge capacity, reduces internal resistance and polarization losses, extends cycle life, and improves lithium-ion battery utilization efficiency. Both excessively high and low densities hinder effective lithium ion intercalation and deintercalation.
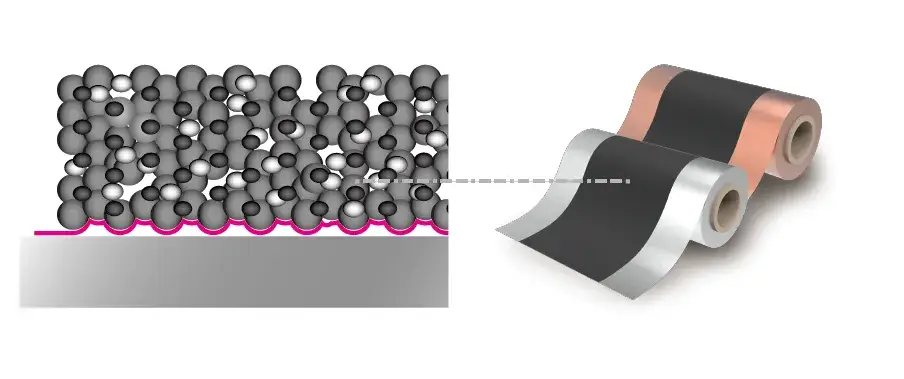
Figure 1. Electrode Sheet Layer
3. Key Factors Influencing Electrode Compaction Density
Four primary factors determine cathode compaction density:
-
Material true density
-
Material morphology
-
Material particle size distribution
-
Electrode manufacturing process
3.1 Material True Density and Its Impact on Electrode Density
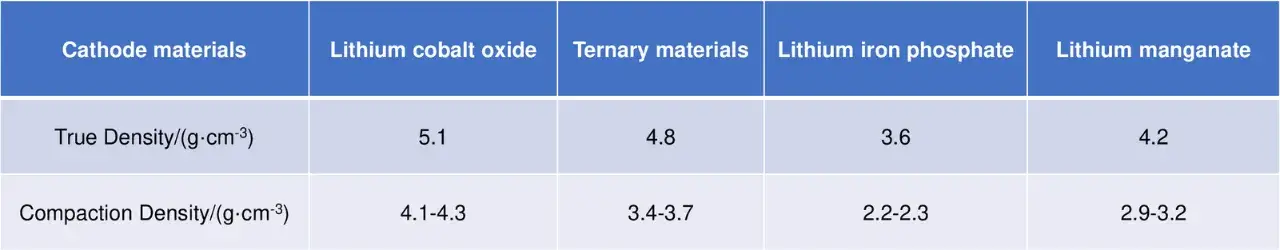
The true density and achievable compaction density of several commercial cathode materials are shown in the table (with the NCM111 material selected for comparison). It is evident that the true densities of these materials follow a consistent pattern: Lithium Cobalt Oxide (LCO) > Ternary Material (NCM111) > Lithium Manganese Oxide (LMO) > Lithium Iron Phosphate (LFP). It should be noted that the true density of different compositions of ternary materials varies with their specific composition.
Table 1. True density and compaction density ranges for commercial cathode materials
3.2 Material Morphology
Material morphology often dominates packing efficiency. For example, commercial LiCoO₂ commonly presents as large primary (single) crystals, while many NCM powders are secondary agglomerates composed of fine primary crystals. Secondary spherical agglomerates inherently contain internal voids. When these secondary particles pack into cathode electrode sheets, inter-spherical voids persist after roll-pressing, lowering electrode density relative to large single crystals.
Practical observation from SEM comparisons shows:
-
Small-primary, tightly agglomerated secondary particles lead to lower compaction density and can shed primary particles during milling and coating.
-
Larger single-crystal particles reduce inter-particle voids and resist detachment, enabling higher electrode density when binders are well dispersed.
These morphology-driven effects explain why two materials with nearly identical true density (e.g., NCM111 vs LiCoO₂) can show notably different electrode densities.

Figure 2. SEM Images of Lithium Cobalt Oxide and Ternary Materials
3.3 Particle Size Distribution
When spheres of equal diameter are packed, significant voids are left between them. If there are no appropriately sized smaller particles to fill these voids, the packing density will be low. Therefore, a suitable particle size distribution can enhance compaction density, whereas an unsuitable distribution can significantly reduce it.
3.4 Electrode Manufacturing Process
Electrode parameters including areal density, binder content, and conductive additive percentage all affect final compaction density. Table 1 shows true densities for common conductive additives and binders.
While material true density imposes fundamental limits, the gap between true density and achievable compaction density suggests significant potential for improvement in materials like NCM.
Table 2. True Density of Common Conductive Agents and Binders

4. Practical Strategies to Increase Cathode Compaction Density
Current approaches focus on three areas: material morphology optimization, particle size distribution control, and electrode process refinement. Examples include developing NCM with LCO-like large single crystals, optimizing NCM particle size distribution, using highly conductive additives to reduce additive percentage, and implementing high-speed mixing for uniform dispersion.
The following case study demonstrates compaction density improvement through NCM morphology and particle size optimization.
4.1 Morphology Optimization
Figure 3 shows SEM images of three NCM variants with different morphologies and their corresponding calendered electrodes. Images (a), (c), and (e) show the pristine powders at identical magnification, while (b), (d), and (f) show lower-magnification SEM of the calendered electrodes.
Material (a) represents conventional NCM morphology – secondary agglomerates of nanocrystals. The corresponding electrode (b) shows large interparticle voids and crushed secondary particles, with some detached primary crystals lacking binder contact.
Material (c) consists of primary single crystals slightly larger than in (a). Electrode (d) reveals minimal interparticle voids. Without secondary particle breakdown issues, crystal detachment is prevented with proper binder distribution.
Material (e), while also agglomerated, features much larger primary crystals with looser inter-crystal contacts. Electrode (f) shows significantly reduced voids, with potential for further improvement through high-speed mixing.
The results for these three morphologies correspond to labels a, c, and e in chart (g). Morphology (a) shows the lowest density, though similar to (c). Morphology (e) achieves significantly higher density, reaching 3.9 g·cm⁻³.

Figure 3. Comparison of NCM morphologies, corresponding electrodes, and compaction densities
4.2 Particle Size Distribution Optimization
Materials with similar D₅₀ values but different D₁₀, D₉₀, D_min, or D_max values exhibit different compaction densities. Both excessively narrow and excessively broad distributions reduce density. Some battery manufacturers specify strict distribution requirements to material suppliers, while others blend products with different distributions to optimize density, as illustrated in Figure 4.

Figure 4. SEM images of cathode electrodes with different particle size distributions
5. Conclusions and Design Guidance
-
Compaction density is a multi-factorial outcome driven by material true density, morphology, particle size distribution, and electrode processing.
-
For NCM-type cathode electrodes, morphology and particle size distribution often offer more room for improvement than changes in true density alone.
-
Balanced optimization — morphology engineering, multimodal particle size distributions, controlled binder/carbon formulation, and calibrated calendaring — delivers the best path to higher electrode density without sacrificing cycle life or electrolyte wetting.
-
Finally, integrate targeted cathode powder battery testing (tapped density, calendered density, SEM, porosity, and electrochemical evaluation) into process qualification to ensure that densification improvements translate to real cell performance gains.
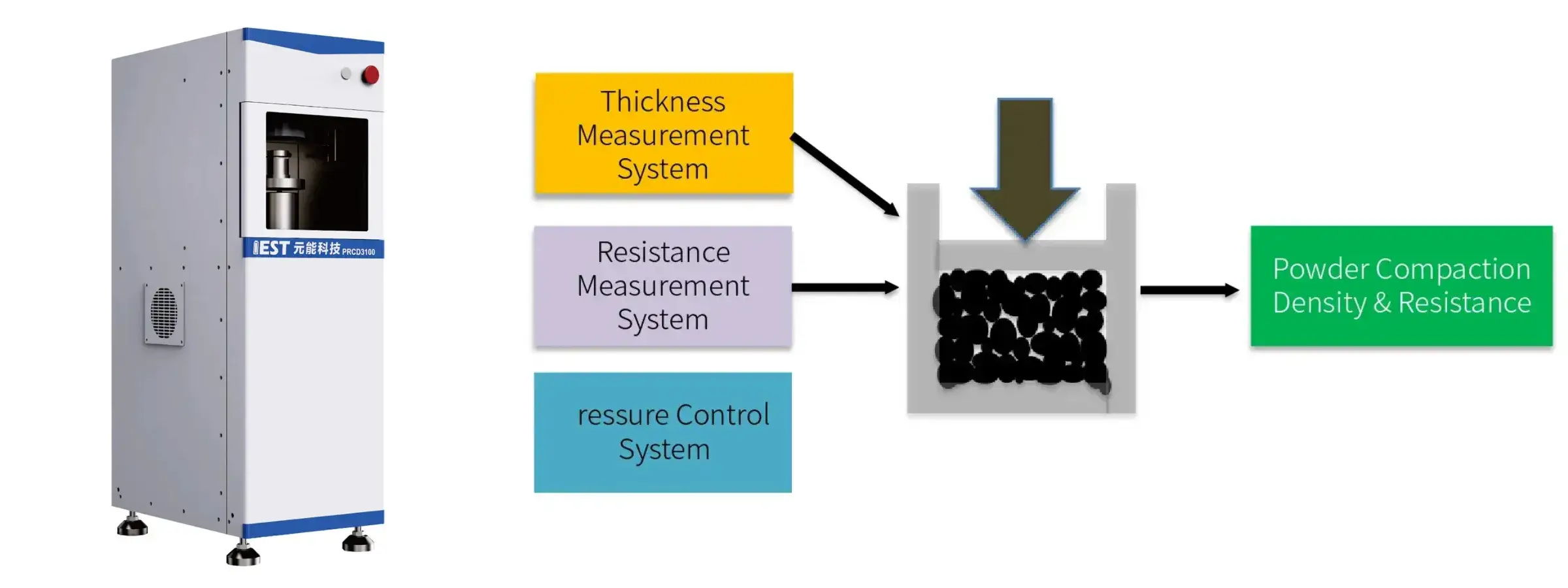
6. IEST Powder Resistivity & Compaction Density Measurement System (PRCD3100) Recommended
PRCD3100 combined with high-precision pressure control,thickness and resistance testing system, free choice of four-probe and two-probe method. Accurately test the resistivity and compaction density of powders for material research and development and batch stability monitoring of powder materials.
Application:
- Lithium (sodium) positive and negative electrode powders (LCO/NCM/LFP/Graphite, etc.),
- Conductive agents
- Solid electrolyte powders
- Other micron-sized powder materials, etc.
Contact Us
If you are interested in our products and want to know more details, please leave a message here, we will reply you as soon as we can.