-
iestinstrument
Advancing Performance in Lithium-Rich Manganese-Based Cathode Materials through Surface Engineering
First Author: Fang Youyou
Corresponding Author: Su Yuefeng, Dong Jinyang, Chen Lai
Publishing Unit: Beijing Institute of Technology, Beijing Institute of Technology Innovation Center
Equipment Used: IEST Powder Resistivity & Compaction Density Measurement System (PRCD1100)
Original Paper: Youyou Fang, Yuefeng Su, Jinyang Dong, Jiayu Zhao, Haoyu Wang, Ning Li, Yun Lu, Yujia Wu, Wenbo Li, Ni Yang, Xiaojuan Wu, Feng Wu, Lai Chen. Ionic‐electronic dual‐conductor interface engineering and architecture design in layered lithium‐rich manganese‐based oxides. Carbon Energy, 2024, e642.
1. Abstract
Li-rich Mn-based layered oxides (LLOs) offer high specific capacity but are hindered by low initial coulombic efficiency (ICE), voltage fade, and poor rate/cycling performance. This study summarizes an ionic-electronic dual-conductor (IEDC) surface-engineering strategy that integrates a graphene conductive framework with an epitaxial spinel Li₄Mn₅O₁₂ layer on Li₁.₂Mn₀.₆Ni₀.₂O₂ particles. Density functional theory (DFT), structural characterization, and electrochemical tests show that the IEDC coating lowers polarization, stabilizes lattice oxygen, accelerates Li⁺/electron transport, and markedly improves ICE, rate capability and cycle stability. Key quantitative findings—DFT-predicted Li⁺ migration barrier reduction (0.576 → 0.367 eV) and experimental ICE = 82.9% with 296.7 mAh g⁻¹ initial discharge for the Gr/Li₄Mn₅O₁₂-coated sample—are preserved from the source data.
2. Introduction: Challenges for Li-rich Mn-based cathodes
Li-rich Mn-based layered oxides (LLOs) deliver attractive gravimetric energy because they combine transition-metal redox and lattice-oxygen redox. However, their practical deployment is limited by several interrelated issues: low initial coulombic efficiency (ICE), rapid capacity/voltage fade during cycling, and sluggish kinetics at high rates. These shortcomings arise from structural reconstruction, lattice-oxygen instability, and poor electronic/ionic connectivity at the particle surface and electrode-electrolyte interface (EEI). To address these failure modes concurrently, the authors implemented an IEDC surface-coating architecture that targets both electron and Li⁺ transport while protecting lattice oxygen and the EEI.
3. IEDC Surface Coating and Architecture
Figure 1 schematically illustrates the preparation process and design principle of the IEDC interface engineering. The procedure involves a post-heat treatment and chemical coating process within a graphene (Gr) network, which induces the growth of a spinel heterophase on the surface of the bulk material’s layered structure. Consequently, the LMO particles are encapsulated by a spinel-Gr hybrid layer, forming the final LMOSG sample. The high ionic diffusivity of the epitaxial spinel heterophase enhances Li⁺ diffusion kinetics and surface structural stability. Simultaneously, the superior conductivity of the graphene network reduces delithiation polarization and strengthens the electrode-electrolyte interface. This innovative surface reconstruction design significantly improves cycling and rate capability, offering a promising strategy for enhancing LMO cathode stability.
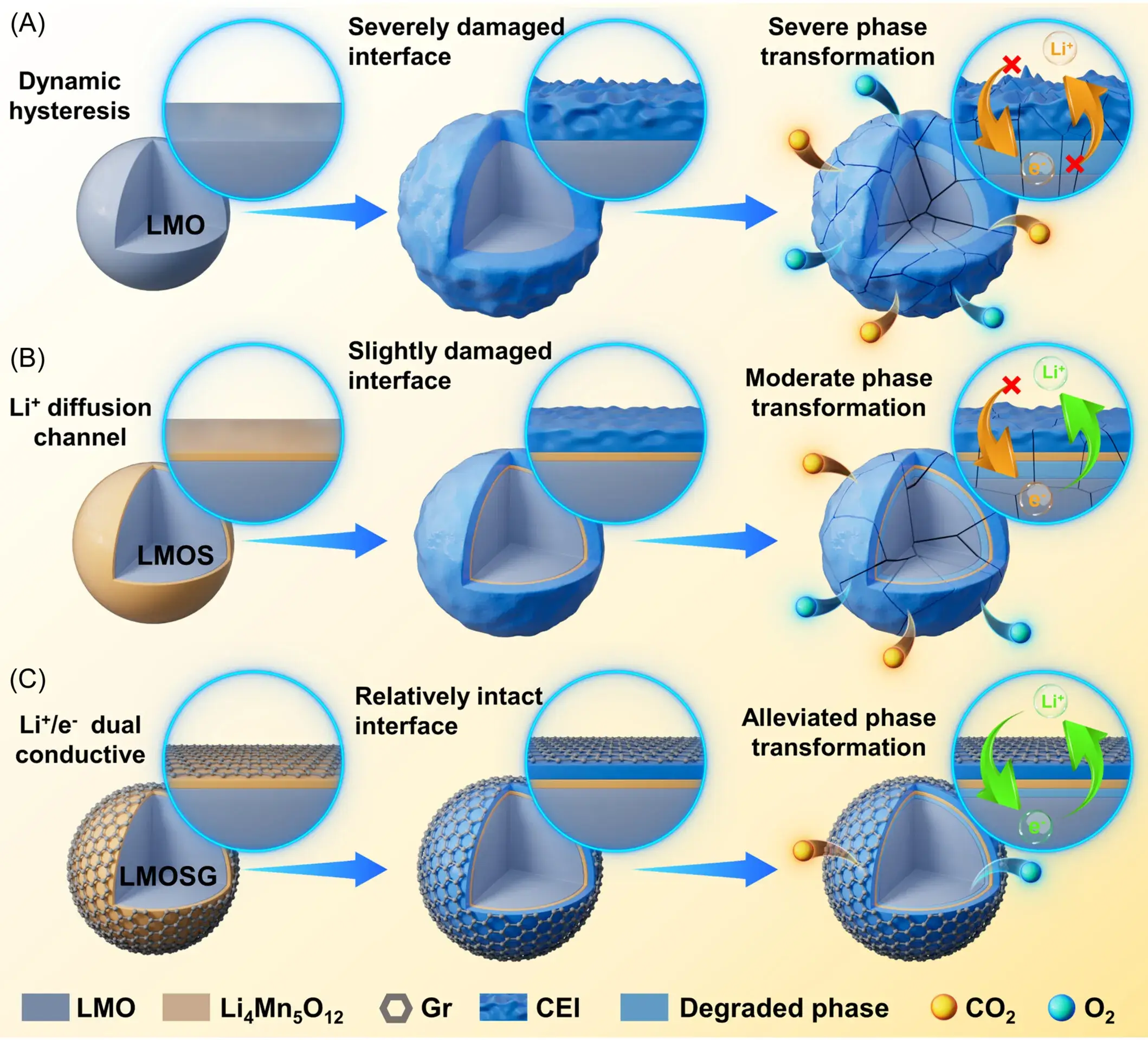
Figure 1. Schematic diagrams of (a) LMO, (b) LMOS, and (c) LMOSG, showing CEI evolution and electron/Li⁺ conduction pathways during electrochemical processes
4. DFT Validation of Enhanced Kinetics and Stability
The effectiveness of IEDC interfacial engineering in promoting Li+ diffusion and stabilizing lattice oxygen is confirmed by mutual validation of density-functional theory (DFT) calculations and experimental results.
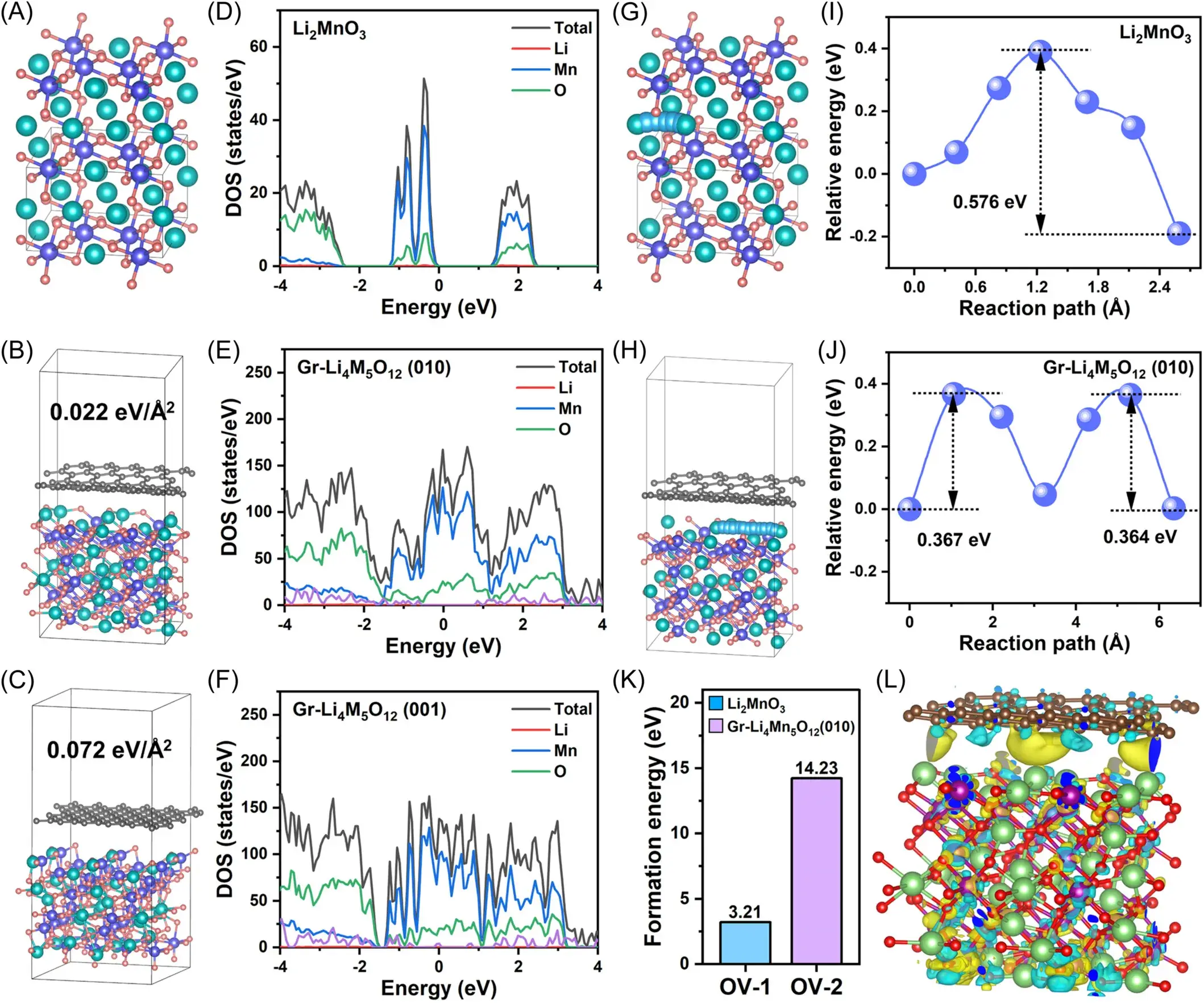
Figure 2. (a)The structure models of (A) Li2MnO3, (B) Gr-Li4Mn5O12(010), and (C) Gr-Li4Mn5O12(001) and the corresponding total density of states plots are illustrated in (D–F). The Li+ diffusion pathways and corresponding diffusion energy barriers of (G, I) Li2MnO3 and (H, J) Gr-Li4Mn5O12(010). (K) The oxygen vacancy formation energy of Li2MnO3 and Gr-Li4Mn5O12(010). (L) The electron density difference for the Gr-Li4Mn5O12(010) surface.
Figure 2 presents structural models and corresponding total density of states (DOS) for Li₂MnO₃, Gr-Li₄Mn₅O₁₂ (010), and Gr-Li₄Mn₅O₁₂ (001). The introduction of Gr and Li₄Mn₅O₁₂ significantly increases the DOS at the Fermi level, indicating enhanced electronic conductivity for Li₂MnO₃. Analysis of Li⁺ diffusion paths and corresponding energy barriers shows that the barrier for Gr-Li₄Mn₅O₁₂ (010) is reduced to 0.367 eV, compared to 0.576 eV for pristine Li₂MnO₃. This reduction is attributed to the three-dimensional lithium diffusion channels provided by the Li₄Mn₅O₁₂ phase. Furthermore, analysis of oxygen vacancy formation energy reveals a significantly higher value for Gr-Li₄Mn₅O₁₂ (010) than for Li₂MnO₃, demonstrating enhanced lattice oxygen stability due to the IEDC interface. Differential charge density diagrams illustrate the electron distribution at the interface between Gr and Li₄Mn₅O₁₂ (010), showing that electrons readily transfer from graphene to the outer oxygen atoms of Li₄Mn₅O₁₂. These findings underscore the efficacy of IEDC interface engineering in facilitating Li⁺ diffusion and stabilizing lattice oxygen, thereby boosting the electrochemical performance of the lithium-rich cathode.
5. Structural and Spectroscopic Evidence of the Surface Coating
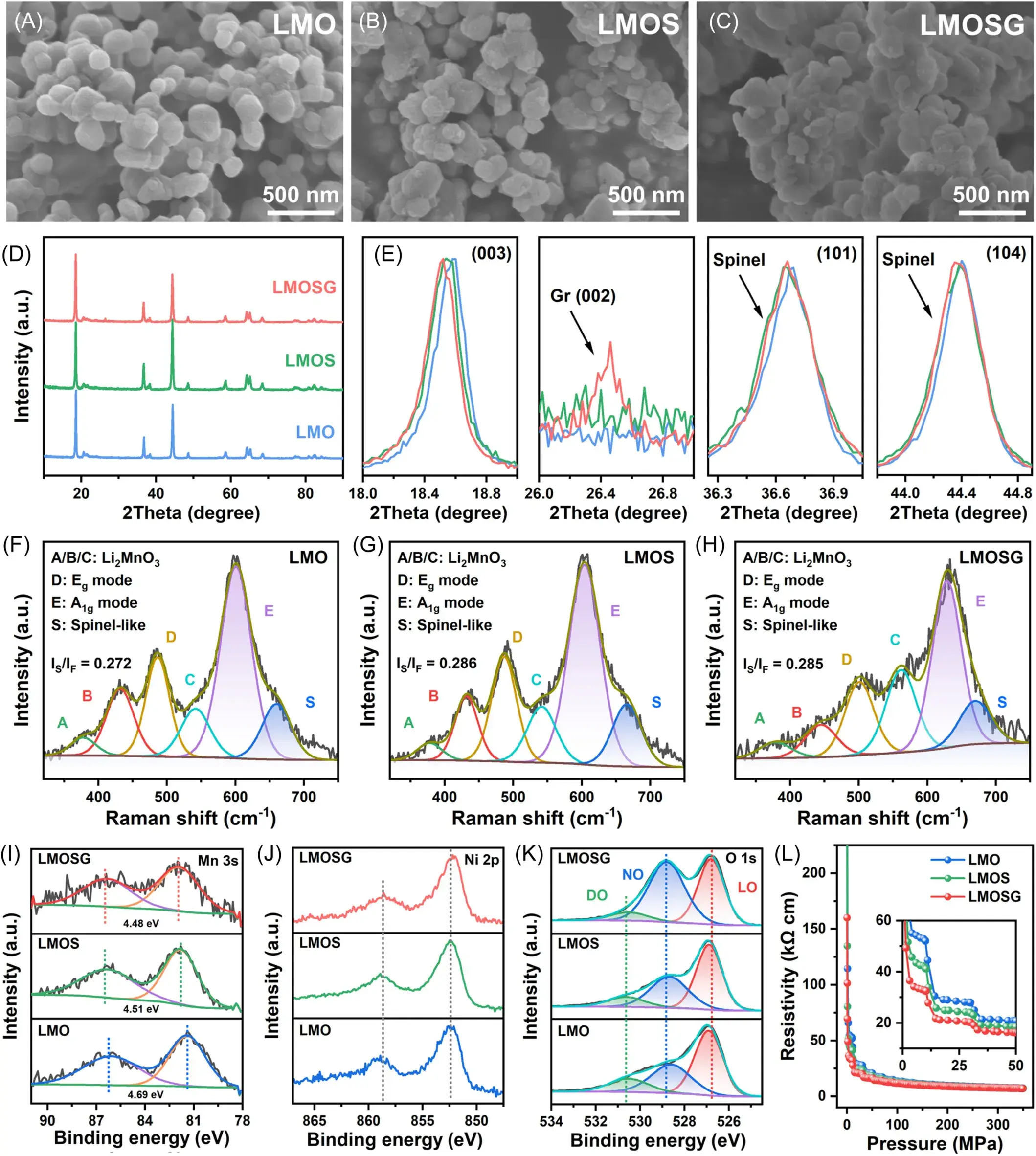
Figure 3. SEM images of the (A) LMO, (B) LMOS, and (C) LMOSG samples. (D) Panoramic XRD patterns with (E) expanded 2θ regions. Raman spectra of the (F) LMO, (G) LMOS, and (H) LMOSG samples. (I) Mn 3s XPS spectra, (J) Ni 2p XPS spectra, (K) O 1s XPS spectra, and (L) pressure versus resistivity plot for all three samples.
6. Electrochemical performance: ICE, Rate Capability and Cycling Stability
The IEDC strategy led to remarkable improvements in key electrochemical metrics for the Li-rich Mn-based, including initial coulombic efficiency, specific discharge capacity, and cycling stability.

Figure 4. (A) Initial charge‒discharge profiles at 0.1 C between 2.0 and 4.8 V. (B) Rate performance between 2.0 and 4.6 V. (C) Cycling performance at 1 C between 2.0 and 4.6 V. (D) Cycling performance at 5 C between 2.0 and 4.6 V. Corresponding charge‒discharge curves at 1 C for the (E) LMO electrode, (F) LMOS electrode, and (G) LMOSG electrode. Corresponding discharged dQ/dV plots of the (H) LMO electrode, (I) LMOS electrode, and (J) LMOSG electrode.
Electrochemical tests compare LMO (base), LMOS (spinel coated), and LMOSG (spinel + graphene) electrodes under identical protocols:
-
Initial Coulombic Efficiency (ICE): LMOSG achieves 82.9% ICE at 0.1 C, which is a notable improvement over unmodified LMO.
-
Initial discharge capacity: At 0.1 C, LMOSG delivers 296.7 mAh g⁻¹.
-
Rate capability: LMOSG sustains superior capacity across 0.1–5 C, retaining 176.5 mAh g⁻¹ at 5 C, while bare LMO delivers 151.8 mAh g⁻¹ at 5 C. This demonstrates improved high-rate kinetics attributable to the IEDC coating.
-
Cycling stability: Under 1 C and 5 C cycling windows (2.0–4.6 V), LMOSG shows higher capacity retention and reduced voltage decay relative to LMO and LMOS—evidence that the coating mitigates degradation mechanisms during prolonged cycling.
-
dQ/dV analysis: Differential capacity curves show smaller shifts and reduced peak attenuation for LMOSG, consistent with lower polarization and more reversible redox behavior.
Collectively, these electrochemical metrics validate that the IEDC surface coating simultaneously raises ICE, enhances rate capability, and stabilizes cycling by optimizing electron/Li⁺ transport and protecting the electrode-electrolyte interface(EEI).
7. Practical implications for Electrode-Electrolyte Interface and Surface Coating Design
The IEDC concept illustrates several transferable design principles for Li-rich Mn-based cathodes:
-
Dual-function coatings: Combine an electronically conductive scaffold (e.g., graphene) with an ion-conductive surface phase (e.g., spinel) to reconcile electrical and ionic constraints without sacrificing active loading.
-
Thin, epitaxial layers: Maintain coating thinness and epitaxial continuity to preserve bulk active material while delivering enhanced surface transport.
-
Stabilize lattice oxygen: Increase oxygen vacancy formation energy at the surface to reduce O₂ release at high voltages.
-
Improve EEI chemistry: A conductive coating that also suppresses electrolyte parasitics reduces irreversible capacity loss during formation and raises ICE.
-
Characterize multi-scale transport: Pair DFT with resistivity-vs-pressure and electrochemical testing to link atomic-scale transport changes to measurable rate capability gains.
Adopting these strategies can accelerate the translation of high-energy LLOs into cells that meet practical ICE and rate requirements.
8. Summary and Outlook
This study successfully applied a straightforward IEDC interface engineering technique to LMO particles, creating a multi-layered surface architecture comprising a conductive Gr framework and a spontaneously formed ion-conductive spinel Li₄Mn₅O₁₂ heteroepitaxial layer. Comprehensive testing verified that this dual-conductor surface reduces polarization, maintains crystal structure stability, and promotes rapid electron/ion diffusion. DFT calculations indicated enhanced electronic conductivity at the Gr/Li₄Mn₅O₁₂ (010) interface compared to the pristine LMO material, attributable to a significantly reduced electronic band gap. Further theoretical investigation revealed a highly interconnected 3D Li⁺ percolation network and lowered Li⁺ migration energy barriers within the layered-spinel interfacial heterostructure. Crystallographic and electrochemical analyses demonstrated that the innovative multi-layer structure suppresses lattice oxygen release and transition metal migration/dissolution, while enhancing ionic conductivity, thereby improving overall electrochemical performance. This effective and meticulously designed surface reconstruction method holds promise for contributing to the development of versatile interfaces for next-generation electrochemical energy storage systems.
9. Original Paper
Youyou Fang, Yuefeng Su, Jinyang Dong, Jiayu Zhao, Haoyu Wang, Ning Li, Yun Lu, Yujia Wu, Wenbo Li, Ni Yang, Xiaojuan Wu, Feng Wu, Lai Chen. Ionic‐electronic dual‐conductor interface engineering and architecture design in layered lithium‐rich manganese‐based oxides. Carbon Energy, 2024, e642.
Contact Us
If you are interested in our products and want to know more details, please leave a message here, we will reply you as soon as we can.