-
iestinstrument
Analysis of Surface Stress Evolution in Graphite Electrodes During Early Cycling
1. Article Abstract
We present a clear, engineering-focused translation of Amartya Mukhopadhyay’s in-situ optical stress study on thin-film graphite electrodes (CVD C) paired with lithium metal. The work documents how irreversible surface stress develops during the first cycles, ties that stress to SEI formation and solvent-co-intercalation, and quantifies how stress scales with voltage, cycle number, and film architecture. These findings provide practical insight for researchers performing lithium-metal testing and interfacial/mechanical characterization of electrode materials.
2. Introduction
Mechanical stress in electrodes couples directly to electrochemical performance. When graphite electrodes expand and contract during lithiation/delithiation, substrate constraints can produce film curvature and net stress. Such stresses influence SEI formation, particle cracking, and long-term electrode integrity — all of which are critical for both lithium-ion and lithium-metal testing contexts. This study uses an in-situ optical deflection method to monitor stress evolution of ultra-thin graphite films and links mechanical observables to electrochemical events.
3. Measurement Principle
A parallel laser beam is focused onto the backside of a quartz substrate, which serves as the carrier for the graphite electrode. During lithiation and delithiation, the rigid substrate constrains the in-plane expansion and contraction of the active film (graphite carbon in this case), causing the substrate/film system to bend. Consequently, the laser beam deflects upon reflection from the substrate. Since the quartz substrate undergoes elastic deformation, the stress in the film is proportional to the induced change in the substrate’s curvature. By monitoring the change in the spot separation of the deflected beam reflected from the substrate’s backside, the in-situ development of stress within the graphite electrode can be tracked.
4. Experimental Details
4.1 Sample information
The graphite carbon film (CVD C) was prepared via Chemical Vapor Deposition (CVD) and graphitization principles onto a 1-inch diameter quartz wafer (250 nm thick). Additionally, a 0.5 nm thick Al₂O₃ layer was deposited via Atomic Layer Deposition (ALD) on some graphite carbon layers. This Al₂O₃ coating was intended to block SEI formation and solvent-cointercalation on the anode
4.2 Experiments
Model cells comprising the CVD C film versus lithium metal were assembled in a custom electrochemical cell. Galvanostatic discharge/charge cycling was performed. Stress changes in the CVD C film, along with its electrochemical behavior, were investigated by monitoring the curvature (bending) of the substrate/film system.
5. Result Analysis
5.1 Electrochemical Behavior During Initial Cycling
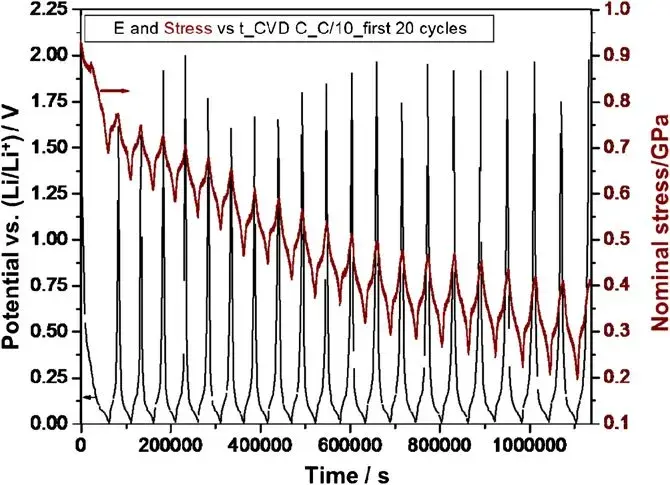
Figure 1. Potential and stress vs. time during lithiation/delithiation cycles for CVD C.
The first cycle showed significant Li capacity insertion during the lithiation half-cycle, while the reversibility during the delithiation half-cycle was relatively low. Apart from the noticeable irreversible capacity loss in the first cycle, most capacity in subsequent cycles was reversible and consistent across different C-rates (C/5, C/10, C/20). The graphite film electrode (CVD C) repeatedly achieved capacities close to the theoretical value at these rates, indicating that Li diffusion within this material is not rate-limiting. Furthermore, after 50 cycles, no capacity fade or significant macro/microstructural damage was observed, and the voltage profiles remained stable.
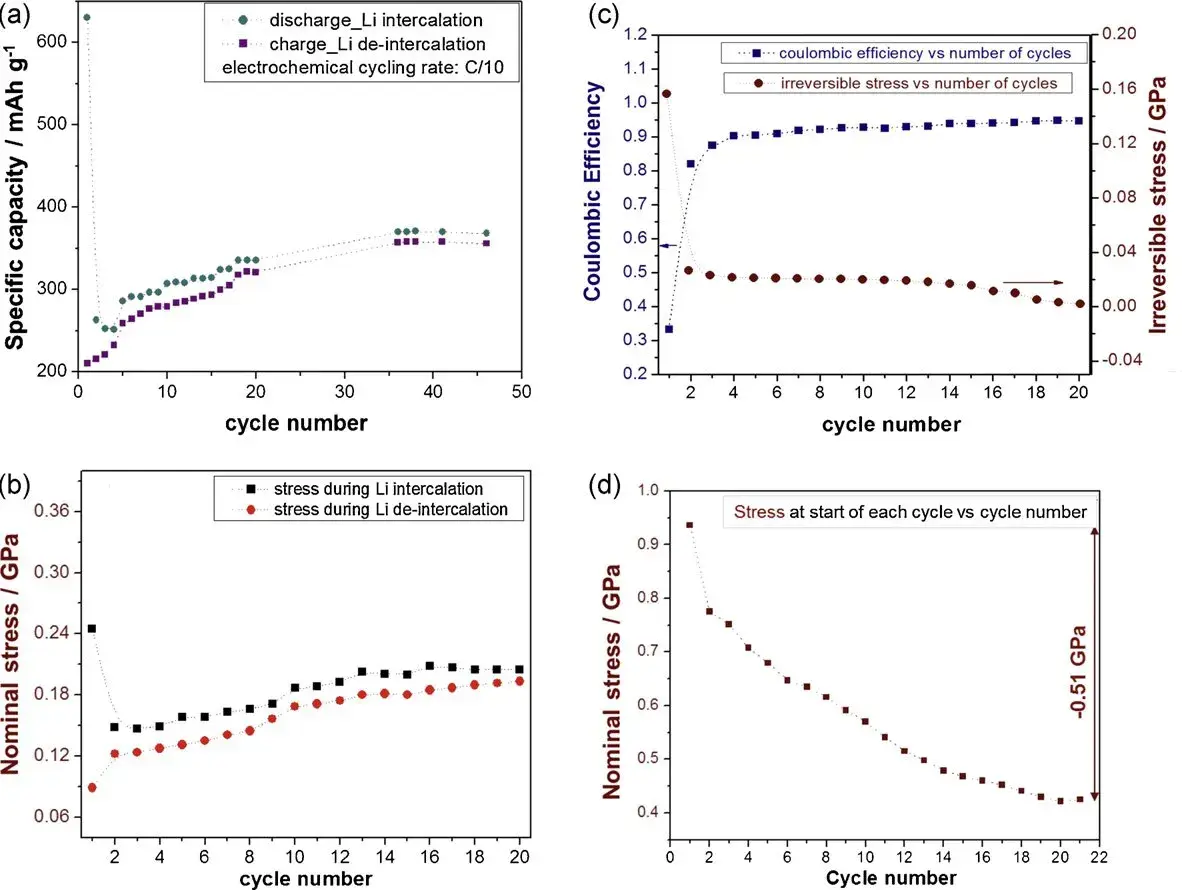
Figure 2. (a) Li capacity vs. cycle number during lithiation and delithiation half-cycles; (b) Nominal stress measured during lithiation and delithiation half-cycles; (c) Coulombic Efficiency (CE) and irreversible stress; (d) Residual stress present in the sample at the start of each cycle.
Irreversible capacity, defined as the difference in Li capacity between lithiation and delithiation, is described by the Coulombic Efficiency (CE). The study reported that the accumulated irreversible stress during initial cycles was up to twice as high as the stress induced by the reversible component. Furthermore, the surface effect (film thickness effect) on irreversible stress indicated that the irreversible component is primarily determined by one or more phenomena occurring near the film surface. The first cycle recorded high capacity and consequent high stress during lithiation, while capacity and stress reversal during delithiation were relatively low, leading to significant differences in both capacity and stress between the first two half-cycles and a very low CE value for the first cycle. The correlation between electrochemical behavior and stress development in the first ~20 cycles, coupled with the finding that irreversible stress originates from surface phenomena, strongly suggests that a significant portion of the irreversible stress is directly related to the formation of the Solid Electrolyte Interphase (SEI) layer.
Stress evolution during the initial cycles is shown in more detail in Figures 2b-2d. The authors used graphite carbon films with lattice parameters aligned parallel to the substrate, ensuring stress development was solely related to changes within the film. Net compressive stress was generated during discharge (lithiation) and subsequently reversed during charge (delithiation). In the first lithiation cycle, a substantial portion of the compressive stress was not reversed. The magnitude of irreversible stress decreased significantly by the second cycle, and this trend continued until the stress difference between subsequent half-cycles became negligible, meaning stress development during delithiation/lithiation became almost fully reversible. The authors posit that this irreversible stress constitutes important feedback for assessing material characteristics.
6. Source Attribution: SEI formation and surface-layer effects on irreversible stress
6.1 Surface Layer Effect on Irreversible Stress
Through hypothesis testing, the authors concluded that the irreversible stress originates from the surface layer of the graphite carbon film, independent of the CVD C film’s bulk thickness. Their thickness variation studies reasoned that if irreversible stress arose from the film’s bulk, the corresponding stress-thickness product would scale with film thickness. Conversely, if the contribution stems from the graphite carbon layer’s surface, the stress-thickness product would be independent of film thickness.
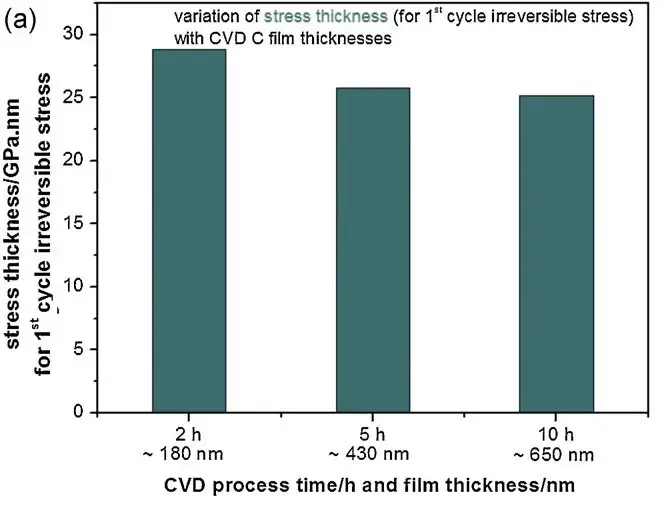
Figure 3. (a) Actual stress-thickness product related to irreversible stress for films of different thicknesses.
6.2 Stress Variation at Different Potentials
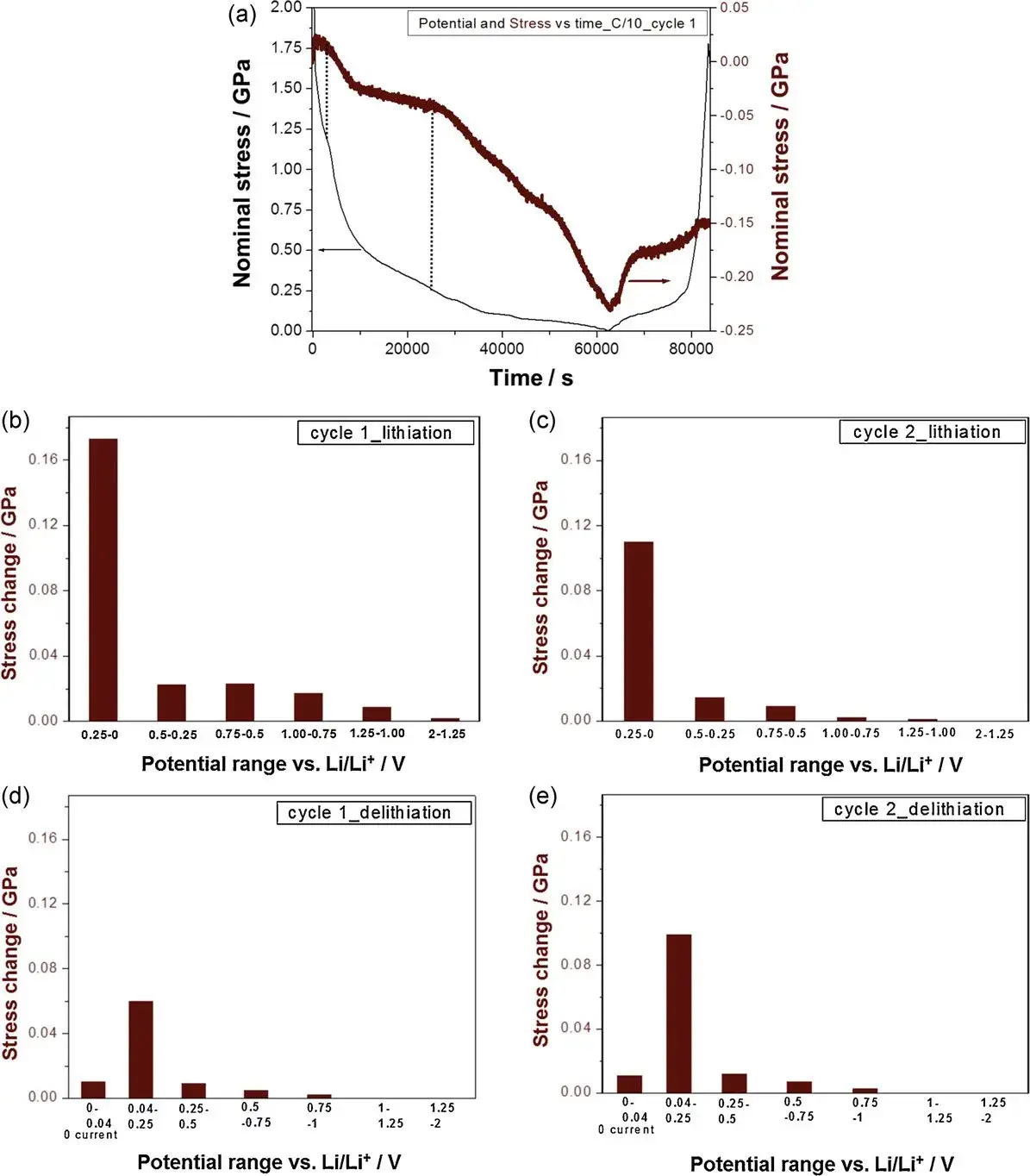
Figure 4. (a) Potential and nominal stress (equivalent to residual stress in the deposited film) vs. time for the first cycle; (b)-(e) Stress evolution during lithiation/delithiation half-cycles within different potential ranges.
It is known that actual Li intercalation primarily occurs below ~0.25 V, where a rapid change in compressive stress is observed from the potential data. Once the potential reaches about 1.0 V, a protective SEI layer forms, almost completely suppressing solvent-cointercalation. Figures 4b-e summarize the stress contributions measured within different voltage ranges during the 1st and 2nd cycle lithiation and delithiation processes. The authors indicate that most of the compressive stress during the lithiation half-cycle occurs below 0.25 V, corresponding to actual Li-ion intercalation. In the initial cycles, this voltage range is also accompanied by ongoing SEI formation, which explains the small amount of irreversible compressive stress still observed after the second cycle. Any stress at higher potentials (>0.25 V) during the first cycle is directly linked to SEI formation or potentially solvent-cointercalation.
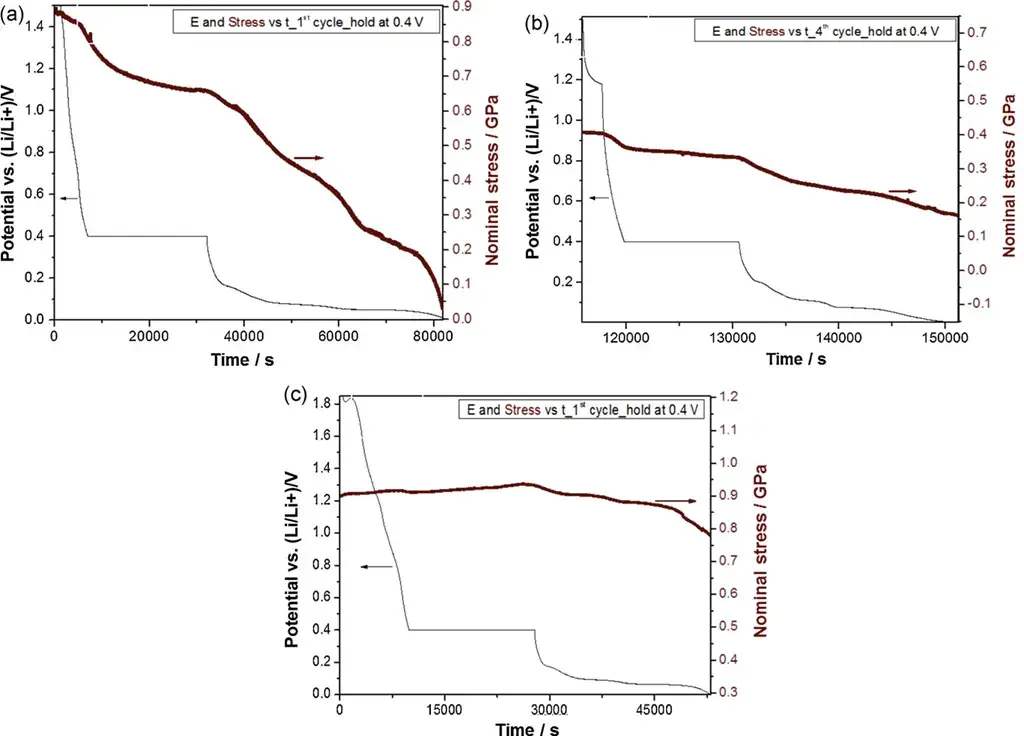
Figure 5. (a) Stress change during the first lithiation half-cycle; (b) Stress change during the second lithiation half-cycle; (c) Stress change during the first lithiation half-cycle for Al₂O₃-coated CVD C film.
Stress changes are attributed to: Figure 5a: Li intercalation + SEI formation + solvent-cointercalation; Figure 5b: Li intercalation + slow, continued passivating SEI formation; Figure 5c: Actual Li intercalation into graphite (SEI formation blocked by Al₂O₃ coating). This further confirms that the primary source of irreversible stress is SEI formation.
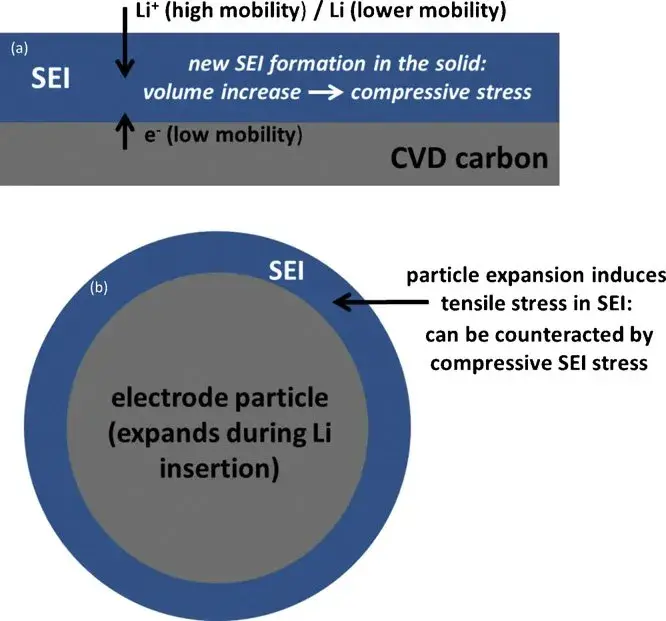
Figure 6. (a) Schematic of SEI film formation on the thin-film graphite electrode and accompanying stress; (b) Expected behavior within a graphite particle and the surrounding SEI layer.
7. Quantitative Relationships: Irreversible vs. Reversible Stress in Early Cycling
Key quantitative outcomes reported in the original study include:
-
The cumulative irreversible stress during the initial cycles was roughly two times larger than the reversible stress component caused by lithiation/delithiation strains.
-
After the first few cycles (≈2–20 half cycles), the irreversible stress magnitude declines substantially and the stress response approaches full reversibility.
These values indicate that the mechanical impact of early SEI formation is both large and primarily confined to a near-surface region of the graphite electrodes.
8. Practical Implications For electrode Design and Testing Protocols
For engineers and scientists testing graphite electrodes or developing negative electrodes for lithium-metal systems, the results suggest several practice-oriented recommendations:
-
Measure in-situ stress during the first cycles. Early irreversible stress is a sensitive proxy for SEI formation and solvent-co-intercalation risk.
-
Consider ultrathin surface coatings (e.g., ALD Al₂O₃) to separate SEI-related surface mechanics from bulk intercalation mechanics during diagnosis.
-
When scaling from thin films to composite electrodes, recognize that surface processes (SEI, binder/solvent interactions) can dominate early mechanical degradation even when the active layer is relatively thin.
These steps help translate mechanistic lab findings into actionable design and qualification criteria for practical electrodes.
9. Summary
This in-situ optical curvature study demonstrates that the largest mechanical penalty for thin-film graphite electrodes occurs during the earliest cycles and originates from near-surface chemical processes—primarily SEI formation and any solvent-assisted co-intercalation. After the initial conditioning cycles, the mechanical response becomes largely reversible. These insights inform both fundamental interfacial studies and practical electrode engineering for graphite electrodes in lithium-ion and lithium-metal testing platforms.
10. References
Mukhopadhyay A, Tokranov A, Xiao X, et al. Stress development due to surface processes in graphite electrodes for Li-ion batteries: A first report[J]. Electrochimica Acta, 2012, 66(none):28-37.
11. IEST Related Test Instruments Recommendations:
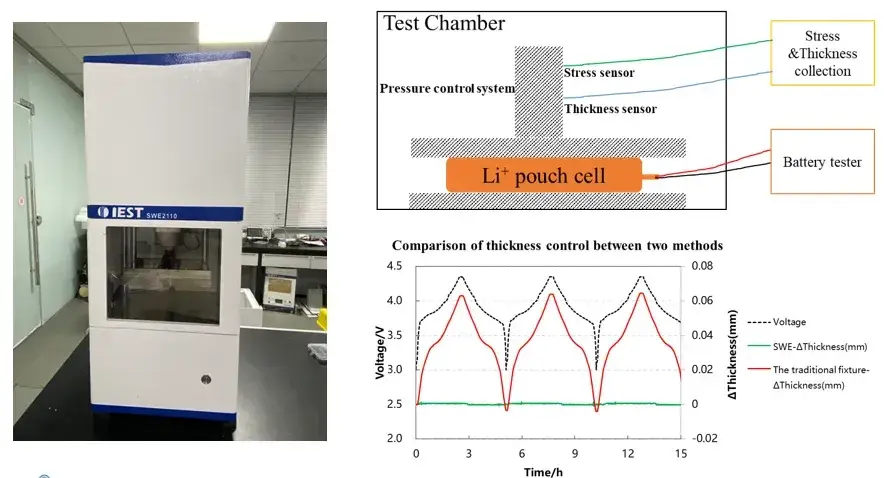
SWE series in-situ swelling analysis system (IEST):
1. Integration of multiple in-situ cell characterization methods (stress & swelling thickness): Measure the swelling thickness and swelling force during the charging and discharging process of the cell at the same time, and quantify the changes in the swelling thickness and swelling force of the cell;
2. More detailed and stable testing system: utilizing a highly stable and reliable automated regulation platform, equipped with high precision thickness measurement sensors and pressure regulation system, the relative thickness measurement resolution is 0.1µm, realizing the long cycle monitoring of the long-term charging and discharging process of the battery cell;
3. Diversified environmental control and testing functions: SWE series equipment can adjust the temperature of the charging and discharging environment, which is helpful for the study of the swelling behavior of the electric core under high and low temperature conditions; in addition to the conventional thickness and pressure testing, it can also realize the testing of parameters such as the swelling force of the electric core, the compression modulus, and the compression rate.
Contact Us
If you are interested in our products and want to know more details, please leave a message here, we will reply you as soon as we can.




